Mineralocorticoid receptor (MR)/NADPH oxidase (NOX) signaling is involved in the development of obesity, insulin resistance, and renal diseases; however, the role of this signaling on steatotic preneoplastic liver lesions is not fully elucidated. We determined the effects of the MR antagonist potassium canrenoate (PC) on MR/NOX signaling in hepatic steatosis and preneoplastic glutathione S-transferase placental form (GST-P)-positive liver foci. Rats were subjected to a two-stage hepatocarcinogenesis model and fed with basal diet or high fat diet (HFD) that was co-administered with PC alone or in combination with the antioxidant alpha-glycosyl isoquercitrin (AGIQ). PC reduced obesity and renal changes (basophilic tubules that expressed MR and p22phox) but did not affect blood glucose tolerance and non-alcoholic fatty liver disease activity score (NAS) in HFD-fed rats. However, the drug increased the area of GST-P-positive liver foci that expressed MR and p22phox as well as increased expression of NOX genes (p22phox, Poldip2, and NOX4). PC in combination with AGIQ had the potential of inhibiting the effects of PC on the area of GST-P-positive liver foci and the effects were associated with increasing expression of an anti-oxidative enzyme (Catalase). The results suggested that MR/NOX signaling might be involved in development of preneoplastic liver foci and renal basophilic changes in HFD-fed rats; however, the impacts of PC were different in each organ.
INTRODUCTION
Potassium canrenoate (PC), a prodrug and major active metabolite of spironolactone (SR), is a diuretic in management of patients with liver cirrhosis and ascites (Bolondi et al., 2006; Angeli et al., 2010). The therapeutic effect on liver cirrhosis and ascites is confirmed in an experimental bile duct ligation model using PC alone (Thiesson et al., 2007) or in combination with furosemide (Jonassen et al., 1998). These effects are mediated by antagonizing mineralocorticoid receptor (MR) (Nasri et al., 2015), which regulates renal water and sodium retention, and potassium secretion. MR activation by either aldosterone or corticosterone leads to inappropriate nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX) activation and accumulation of reactive oxygen species (ROS) (Whaley-Connell et al., 2010). The effects involve several physiological processes (i.e., inflammation, fibrosis, and endothelial dysfunction) in different tissues, including kidney, heart, vasculature, brain, and adipose tissue (Zennaro et al., 2009) and result in abnormal insulin signaling (insulin resistance) (Whaley-Connell et al., 2010). Insulin resistance is accompanied by obesity and liver steatosis; however, the mechanisms of MR/NOX signaling leading to liver steatosis remain uncertain.
Nonalcoholic fatty liver disease (NAFLD) is increasingly recognized as the hepatic manifestation of insulin resistance and the systemic complex known as metabolic syndrome. A population of subjects progresses to a severe form, nonalcoholic steatohepatitis (NASH), and a minority of affected patients eventually develop hepatocellular carcinoma (HCC) (Dongiovanni et al., 2014). The mechanism(s) of these sequential diseases are not clearly understood; however, steatosis-mediated cellular stress and subsequent inflammation might be involved in the pathogenesis (Liu et al., 2016). ROS are considered as one of the key players in tissue injury, and NOX is one of the most important contributors to oxidant generation in several tissues, including liver (Di Meo et al., 2016). NOX plays a role in the development of NAFLD and NASH, and knockout of the NOX subunit can prevent the severity of the diseases in mice (Kono et al., 2000; Chatterjee et al., 2013). Lately, Nie et al. (2015) reported that MR affects cell proliferation, cell cycle processes, and apoptosis in HCC cell lines. However, MR/NOX signaling has not been fully elucidated in liver models of NAFLD and NASH, including development of preneoplastic lesions.
We recently developed a model for steatosis-related early hepatocarcinogenesis using a medium-term liver assay in high fat diet (HFD)-fed rats (Yoshida et al., 2017). In this study, we determined the effects of PC on obesity, insulin tolerance, and renal and liver lesions, and examined steatosis-related preneoplastic liver foci, with hepatic gene expression associated with lipid metabolism and oxidative stress, including NOX. The goal was to determine the impact of MR/NOX signaling on the liver lesions, including preneoplastic lesions.
MATERIALS AND METHODS
Chemicals
N-diethylnitrosamine (DEN; CAS No. 55-18-5, purity > 99%) and PC (CAS No. 2181-04-6, purity > 98%) were purchased from Tokyo Kasei Kogyo (Tokyo, Japan) and Sigma-Aldrich Co., LLC, St. Louis, MO, USA, respectively. Alpha-glycosyl isoquercitrin (AGIQ) (purity > 97%) was provided by San-Ei Gen F.F.I., Inc. (Osaka, Japan).
Animals and treatment
A total of 54 5-week-old male F344/N rats were purchased from Japan SLC, Inc. (Shizuoka, Japan), maintained in an air-conditioned room (room temperature, 22 ± 3°C; relative humidity, 56 ± 11%; 12-hr light/dark cycle), and given free access to a powdered diet (Oriental MF; Oriental Yeast, Tokyo, Japan) and tap water. After a 1-week acclimatization period, a medium-term liver carcinogenesis bioassay (Ito et al., 2003) was regardfully conducted using the following procedure. All animals received an intraperitoneal injection of DEN at a dose of 200 mg/kg body weight and were randomly divided into four groups. The control group (CTL) received normal diet, and all others received HFD (D12451; Research Diets, Lane, NJ, USA) containing 0 (HFD group) or 300 ppm PC (HFD+PC group) for 9 weeks starting at 2 weeks after DEN initiation. PC at a dose of 300 ppm (correspondent to 50 mg/kg/day) was administered as previously reported (Bos et al., 2004). The fourth group was administered HFD and PC as well as 5,000 ppm AGIQ in the diet (HFD+PC+AGIQ group) (Nyska et al., 2016). All the animals were subjected to two-thirds partial hepatectomy 1 week after PC and/or AGIQ treatment to develop preneoplastic liver lesions. Animals were observed clinically during the study. Glucose tolerance test was conducted in rats (n = 4 or 5 per group) after overnight fasting between 10 and 11 weeks of study as described previously (Catalogna et al., 2016), and blood glucose levels were measured during 0-120 min after administration of glucose solution (2.0 g/kg) using a blood glucose meter GT-1830 (ARKRAY, Inc., Tokyo, Japan). At the end of the 9-week experiment, the rats were euthanized by exsanguination under isoflurane anesthesia with overnight fasting. The livers, kidneys, and abdominal adipose tissues (surrounding the spermatic cord) were excised and weighed, and the sliced samples were fixed in 4% paraformaldehyde in 0.1 M phosphate-buffered formalin (pH 7.4; Wako Pure Chemicals Industries, Ltd., Osaka, Japan) for histopathology and immunohistochemistry. The liver pieces were frozen in liquid nitrogen and stored at −80°C until further analysis. All procedures in this study were conducted in compliance with the Guidelines for Proper Conduct of Animal Experiments (Science Council of Japan, June 1, 2006), and the protocol was approved by the Animal Care and Use Committee of the Tokyo University of Agriculture and Technology.
Reverse transcription polymerase chain reaction (RT-PCR) analysis
Analysis of mRNA levels of the genes listed in Supplemental Table 1 in liver tissues (n = 6 animals per group) was performed using RT-PCR. Total RNA was extracted using RNeasy® Mini kit (Qiagen, Hilden, Germany), according to the manufacturer’s instructions. First-strand cDNA was synthesized from 2 μg total RNA in a 20-μL total reaction mixture containing dithiothreitol, deoxynucleoside triphosphates, random primers, RNaseOUT, and SuperScriptTMIII Reverse Transcriptase (Life Technologies, Carlsbad, CA, USA). RT-PCR was performed using the SYBR® Green PCR Master Mix (Life Technologies) and the StepOnePlusTM Real-Time PCR System (Life Technologies), according to the manufacturer’s protocol. The PCR primers were designed using the Primer Express software (Version 3.0; Life Technologies). The relative differences in gene expression were calculated using threshold cycle (Ct) values that were first normalized to those of the hypoxanthine phosphoribosyl transferase 1 (Hprt) gene, the endogenous control in the same sample, and relative to a control Ct value using the 2−ΔΔCTmethod (Livak and Schmittgen, 2001).
Histopathology and immunohistochemistry
The liver and kidney slices were dehydrated in graded ethanol, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E) for histopathological examination. The hepatic pathological changes, including steatosis, ballooning change, and inflammatory foci, were graded by the NAFLD activity score (NAS) (Kleiner et al., 2005): The H&E-stained specimens were evaluated by scoring the extent of celluar changes based on steatosis (0, < 5%; 1, 5%-33%; 2, 33%-66%; and 3, > 66%), ballooning change (0, none; 1, few balloon cells; and 2, many), and inflammatory foci (0, no foci; 1, < 2 foci; 2, 2-4 foci; and 3, > 4 foci). Renal basophilic tubules were graded by the severity grading method of chronic nephropathy (Ozaki et al., 2001): The H&E-stained specimens were evaluated by scoring the extent of basophilic changes (1, < 25%; 2, 26%-50%; 3, 51%-75%; and 4, > 76%). Immunohistochemical staining was performed with antibodies against GST-P, Ki-67, cleaved (active) caspase-3, MR, p22phox, and adipophilin listed in Supplemental Table 2. The deparaffinized liver and kidney sections were treated with 0.3% H2O2 in methanol for 30 min, and antigen retrieval was performed if necessary. The sections were incubated with primary antibodies overnight at 4°C. An avidin-biotin-peroxidase complex method with VECTASTAIN® Elite ABC kit (Vector Laboratories Inc., Burlingame, CA, USA) was employed with 3,3’-diaminobenzidine as a chromogen, followed by light hematoxylin staining. The numbers and areas of GST-P-positive foci (> 0.2-mm diameter) and the total areas of the liver sections were quantified using Scion Image (Scion Corp., Frederick, MD, USA) (Hara et al., 2014). Ki-67- and active-caspase-3-positive cells were examined in a total of more than 1,000 cells within GST-P-positive foci (inside foci) per animal. Ki-67- and active-caspase-3-positive cells were examined in a total of more than 300 cells in non-GST-P-positive foci (outside foci) per animal.
Statistical analysis
All data were expressed as mean + standard deviation. Significance of the differences among groups was determined using the Tukey or Steel-Dwass multiple comparison test. A p-value < 0.05 was considered statistically significant.
RESULTS
Treatment with PC alone or in combination with AGIQ decreased the adipose tissue weight in HFD-fed rats
The HFD significantly increased final body weight compared to the CTL group. The treatment with PC alone or in combination with AGIQ significantly decreased final body weight compared to the HFD group (Table 1). Food and water intake significantly decreased with the HFD feeding, independent of treatment with PC with or without AGIQ. Water intake versus the HFD group also significantly decreased with the treatment with PC with or without AGIQ. The HFD with or without PC alone or in combination with AGIQ significantly increased absolute and relative adipose tissue weights, and significantly decreased absolute and relative liver and kidney weights, when compared to the CTL group. In the HFD+PC group, the relative kidney weight significantly decreased compared to the CTL group but increased compared to the HFD group.
Table 1. Final body weight, organ weight, food intake, and water intake in rats after DEN initiation§.
| Group | CTL | HFD | HFD+PC | HFD+PC+AGIQ | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of animals | 13 | 13 | 13 | 15 | ||||||||
| Final body weight (g) | 299.8 | ± | 13.1 | 329.5 | ± | 13.6a | 310.0 | ± | 15.1b | 313.4 | ± | 15.3b |
| Food intake (g/rat/day) | 16.7 | ± | 2.3 | 11.8 | ± | 1.5a | 11.9 | ± | 1.6a | 11.8 | ± | 1.9a |
| Water intake (g/rat/day) | 22.8 | ± | 1.4 | 17.9 | ± | 1.0a | 14.0 | ± | 1.2a,b | 14.3 | ± | 1.0a,b |
| Absolute adipose tissue weight (g) | 7.8 | ± | 0.9 | 12.4 | ± | 1.5a | 11.1 | ± | 1.3a | 11.0 | ± | 1.8a |
| Relative adipose tissue weight (%BW) | 2.6 | ± | 0.2 | 3.7 | ± | 0.3a | 3.6 | ± | 0.4a | 3.5 | ± | 0.4a |
| Absolute liver weight (g) | 7.9 | ± | 0.7 | 6.8 | ± | 0.6a | 6.5 | ± | 0.6a | 6.8 | ± | 0.6a |
| Relative liver weight (%BW) | 2.6 | ± | 0.1 | 2.0 | ± | 0.1a | 2.1 | ± | 0.1a | 2.2 | ± | 0.2a |
| Absolute kidney weight (g) | 2.1 | ± | 0.1 | 1.9 | ± | 0.1a | 2.0 | ± | 0.1 | 1.9 | ± | 0.1a |
| Relative kidney weight (%BW) | 0.684 | ± | 0.025 | 0.586 | ± | 0.025a | 0.631 | ± | 0.028a,b | 0.603 | ± | 0.037a |
Abbreviations: AGIQ, alpha-glycosyl isoquercitrin; BW, body weight; CTL, control; DEN, N-diethylnitrosamine; HFD, high fat diet; PC, potassium canrenoate. §: All animals were subjected to two-thirds partial hepatectomy at week 1 after starting PC treatment with or without AGIQ. Data are shown as the mean ± standard deviation. a: p < 0.05 vs CTL (Tukey’s or Steel-Dwass test). b: p < 0.05 vs HFD (Tukey’s or Steel-Dwass test).
Treatment with PC alone or in combination with AGIQ did not alter blood glucose levels in glucose tolerance test in HFD-fed rats
The HFD with or without PC alone or in combination with AGIQ increased plasma glucose levels in a time-dependent manner compared to the CTL group, and the levels reached maximal levels at 30 min after glucose administration (Table 2). The levels decreased at 60 min and thereafter in all groups. Significant differences were detected at 90 min in the HFD and HFD+PC groups, and at 120 min in the HFD, HFD+PC, and HFD+PC+AGIQ groups compared to the CTL group.
Table 2. Glucose tolerance test in rats after DEN initiation§.
| Group | CTL | HFD | HFD+PC | HFD+PC+AGIQ | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of animals | 5 | 4 | 5 | 4 | ||||||||
| 0‡ | 144.6 | ± | 25.4 | 112.8 | ± | 15.0 | 114.6 | ± | 12.2 | 131.5 | ± | 14.8 |
| 10 | 224.0 | ± | 33.6 | 241.3 | ± | 80.3 | 275.8 | ± | 38.7 | 299.0 | ± | 61.9 |
| 20 | 262.6 | ± | 35.7 | 278.5 | ± | 73.4 | 319.2 | ± | 44.3 | 315.0 | ± | 26.0 |
| 30 | 286.0 | ± | 38.5 | 356.5 | ± | 57.6 | 360.0 | ± | 60.0 | 365.5 | ± | 48.8 |
| 60 | 219.0 | ± | 69.7 | 306.5 | ± | 18.6 | 321.6 | ± | 30.7 | 278.3 | ± | 11.3 |
| 90 | 119.6 | ± | 16.9 | 194.8 | ± | 33.1a | 233.0 | ± | 34.1a | 211.0 | ± | 54.0 |
| 120 | 99.4 | ± | 22.6 | 147.3 | ± | 6.4a | 147.8 | ± | 23.8a | 133.3 | ± | 9.9a |
Abbreviations: AGIQ, alpha-glycosyl isoquercitrin; CTL, control; DEN, N-diethylnitrosamine; HFD, high fat diet; PC, potassium canrenoate. §: All animals were subjected to two-thirds partial hepatectomy at week 1 after starting PC treatment with or without AGIQ. ‡: Post-stimulation time (min). Data are shown as the mean ± standard deviation (mg/dL). a: p < 0.05 vs CTL (Tukey’s or Steel-Dwass test).
Treatment with PC alone or in combination with AGIQ altered the mRNA levels of oxidative stress, antioxidant enzymes, lipogenic enzymes, and transcriptional factors
Regarding lipid metabolism-related genes, Pnpla2 expression significantly increased in the HFD+PC group and the HFD+PC+AGIQ group compared to the CTL group, and significantly increased in the HFD+PC group compared to the HFD group (Fig. 1A). Pparg expression significantly increased in the HFD group, the HFD+PC group and the HFD+PC+AGIQ group compared to the CTL group and Acox1 expression significantly increased in the HFD group compared to the CTL group.
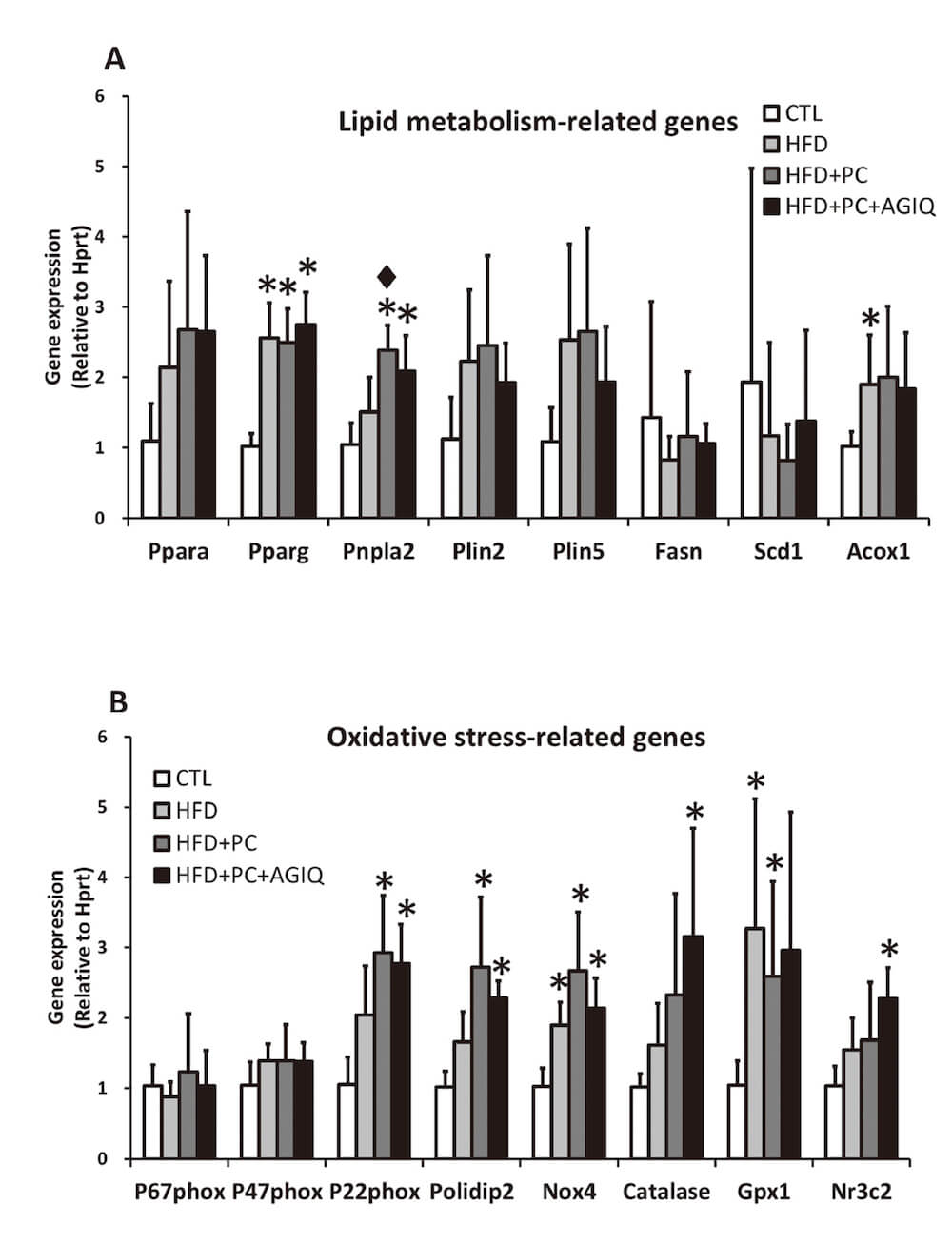
Fig. 1 Hepatic gene expression analysis in rats. The mRNA levels of target genes were measured in liver samples in rats fed with basal diet (CTL) and high fat diet (HFD) co-administered with potassium canrenoate (HFD+PC) and/or alpha-glycosyl isoquercitrin (HFD+PC+AGIQ) by performing real-time reverse transcription-polymerase chain reaction (RT-PCR) normalized to the levels of Hprt1 mRNA. (A) Lipid metabolism-related genes. (B) Oxidative stress-related genes. Abbreviations: Aox1, Alternative oxidase 1; Fasn, Fatty acid synthase; Gpx1, Glutathione peroxidase 1; Nox4, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4; Nr3c2, Nuclear receptor subfamily 3 group C member 2; Plin2, Perilipin 2; Plin5, Perilipin 5; Pnpla2, Patatin-like phospholipase domain containing 2; Poldip2, Polymerase (DNA-directed), delta interacting protein 2; Ppara, Peroxisome proliferator activated receptor alpha; Pparg, Peroxisome proliferator activated receptor alpha; Scd1, Stearoyl-coenzyme A desaturase 1; Hprt1, Hypoxanthine phosphoribosyl transferase 1. *,♦ p < 0.05 vs. CTL or HFD, respectively (Tukey’s or Steel-Dwass multiple comparison test).
Regarding oxidative stress-related parameters, NOX4 expression significantly increased in the HFD group compared to the CTL group, and P22phox, Polidp2, and NOX4 expression significantly increased in the HFD+PC group and HFD+PC+AGIQ group compared to the CTL group (Fig. 1B). Catalase expression significantly increased in the HFD+PC+AGIQ group compared to the CTL group. Gpx1 expression significantly increased in the HFD group and HFD+PC group compared to the CTL group.
A MR target gene, Nr3c2 expression significantly increased in the he HFD+PC+AGIQ group compared to the CTL group (Fig. 1B).
Treatment with PC alone or in combination with AGIQ reduced renal basophilic tubules in HFD-fed rats
Basophilic tubules were scattered in the cortex in rats (Fig. 2A-C), and immunopositivity to MR in the cytoplasms and nuclei was detected in the distal renal tubules and collecting ducts in kidney tissues of each group. An immunopositive reaction to MR and p22phox was also found in basophilic tubules (Fig. 2D, E), and nuclear localization of MR was seen in some cells in basophilic tubules. The score of basophilic tubules significantly increased in the HFD group compared to the CTL group; however, no significant changes were detected in the HFD+PC and HFD+PC+AGIQ groups compared to the CTL group (Fig. 2F).
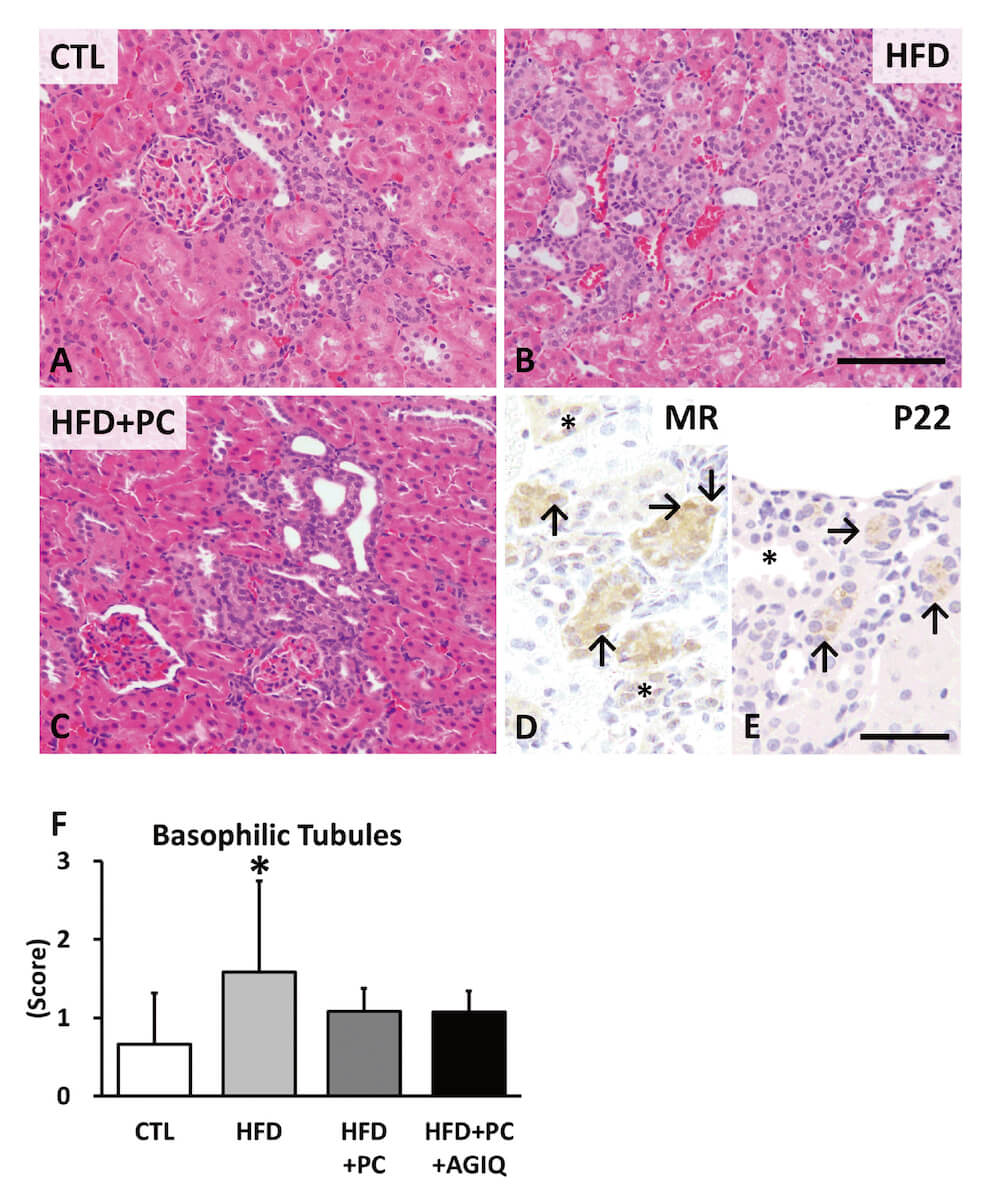
Fig. 2
Histopathological changes in the kidney of rats fed with basal diet (CTL) or high fat diet (HFD) co-administered with potassium canrenoate (HFD+PC) and/or an antioxidant alpha-glycosyl isoquercitrin (HFD+PC+AGIQ). Representative images (A-E) and scores (F) of basophilic tubules in each group. Columns represent mean and standard deviation. (F) * p < 0.05 vs. CTL (Tukey’s or Steel-Dwass multiple comparison test). HE (A-C), Immunohistochemistry of mineralocorticoid receptor (MR) (D) and p22phox (E). (D, F) * Distal tubule; Arrows, basophilic tubule (D, Arrows indicate nuclear localization of MR). Bar = 100 μm (A-C), 40 μm (D, E)
Treatment with PC alone or in combination with AGIQ reduced ballooning change of hepatocytes, while treatment with PC alone increased preneoplastic liver lesions in HFD-fed rats
Fasting decreased glycogen deposition in the hepatocytes in each group, but steatosis was persistently observed with the HFD feeding (Fig. 3A-D), when compared with the liver in our previous study using non-fasting rats (Yoshida et al., 2017). NAS and the scores of steatosis and ballooning change significantly increased in the HFD group with or without PC alone or in combination with AGIQ compared to the CTL group (Fig. 3E-G). The score of ballooning change significantly decreased in the HFD+PC+AGIQ group compared to the HFD group. There were no significant changes in the score of inflammatory foci in any groups (Fig. 3H).
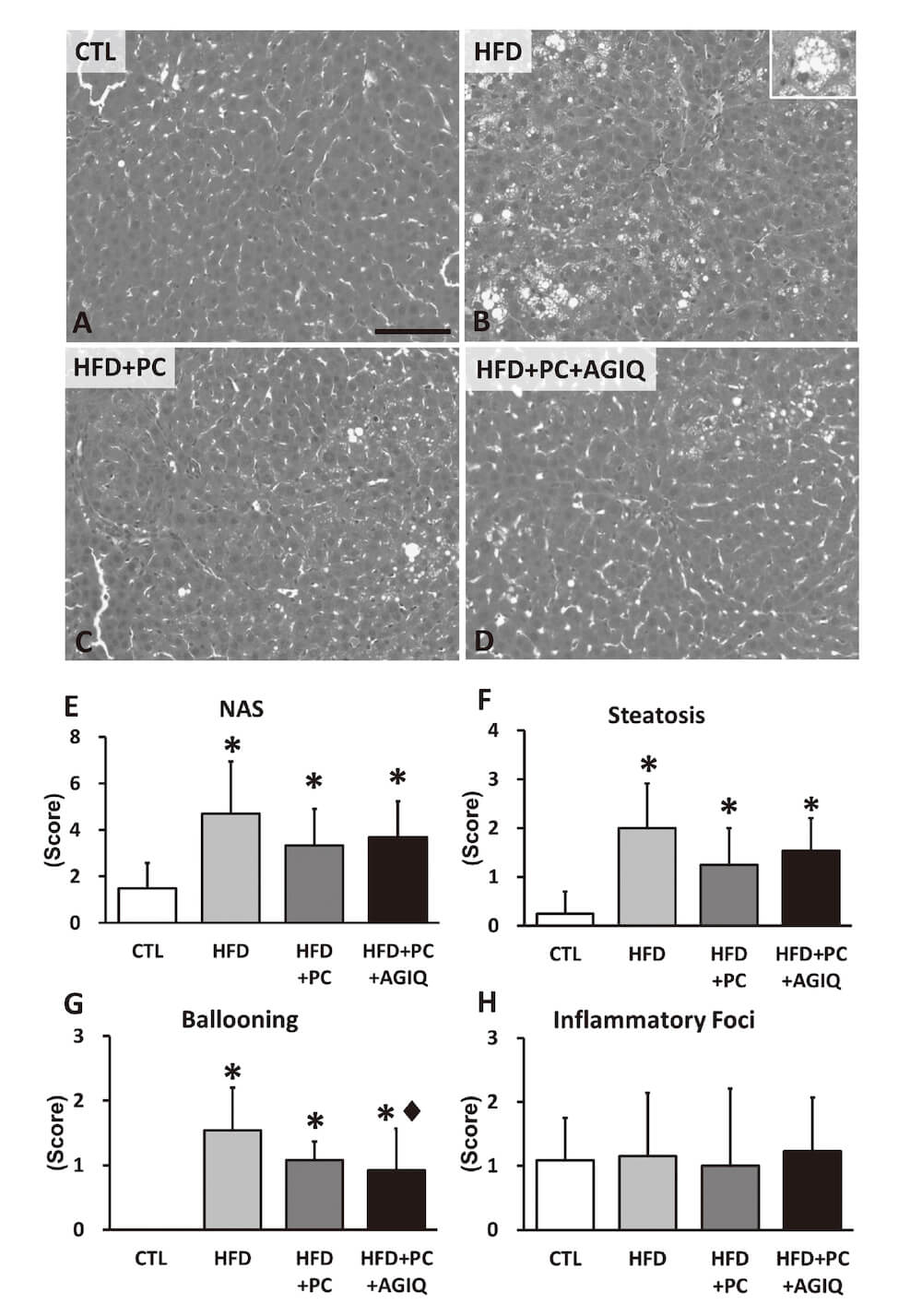
Fig. 3
Histopathological changes in the liver of rats fed with basal diet (CTL) or high fat diet (HFD) co-administered with potassium canrenoate (HFD+PC) and/or an antioxidant alpha-glycosyl isoquercitrin (HFD+PC+AGIQ). Representative images (A-D) and scores of nonalcoholic fatty liver disease (NAFLD) activity score (NAS) (E), steatosis (F), ballooning change (G), and inflammatory foci (H) in each group. Columns represent mean and standard deviation. *,♦ p < 0.05 vs. CTL or HFD, respectively (Tukey’s or Steel-Dwass multiple comparison test). Bar = 25 μm (A-D)
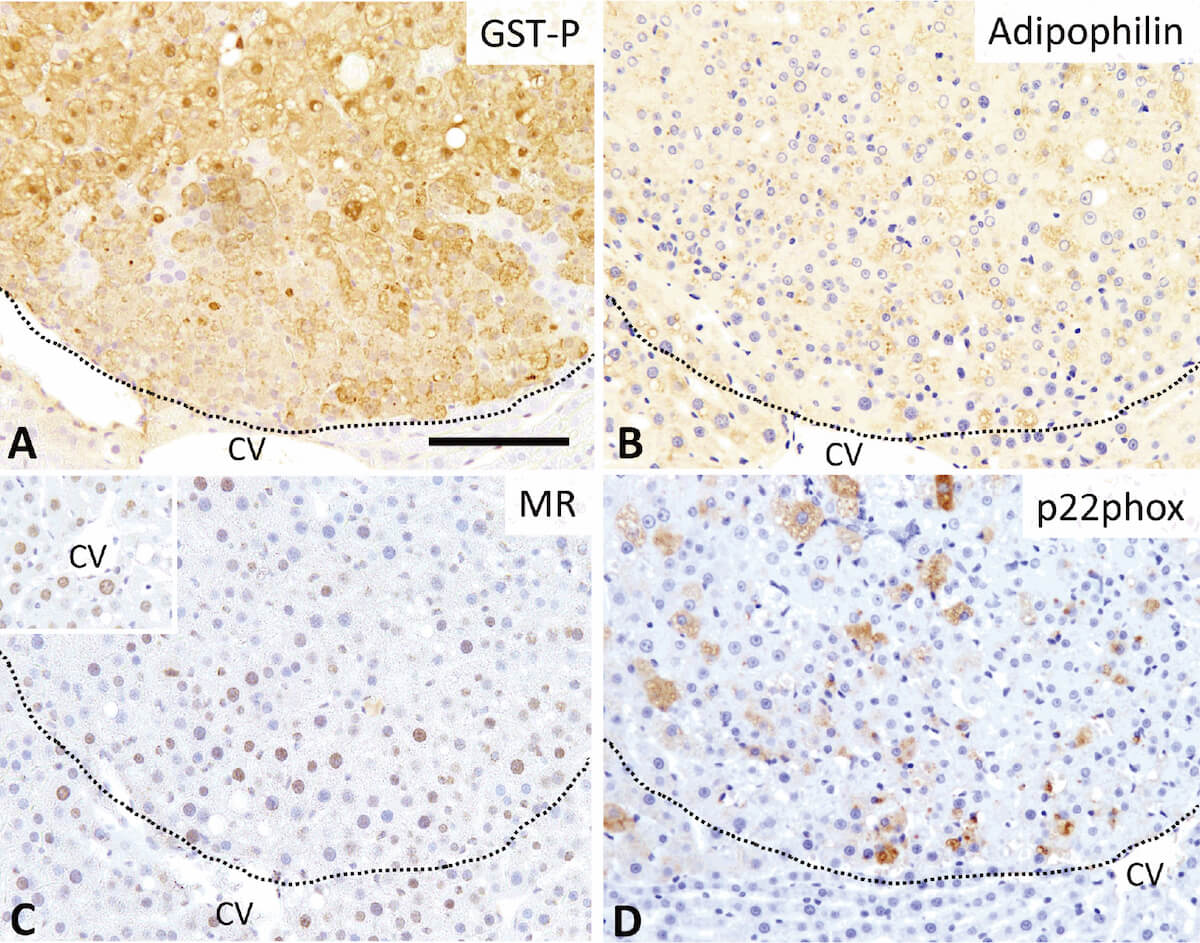
Hepatocytes within liver foci were diffusely immunopositive to GST-P (Fig. 4A), adipophilin (Fig. 4B), and MR (Fig. 4C), and they partially tested positive to p22phox (Fig. 4D). Hepatocytes outside liver foci were strongly immunopositive to adipophilin, especially at the margins of intracytoplasmic lipid droplets. Centrilobular hepatocytes tested positive to MR; nuclear localization of MR was also seen. p22phox-positive hepatocytes were rarely found throughout the liver tissues. The area but not the number of GST-P-positive liver foci was significantly higher in the HFD +PC group than that in the CTL group (Fig. 4E, F). There were no significant differences in Ki-67 and active caspase-3 labeling index in the liver foci among treated groups (Table 3).
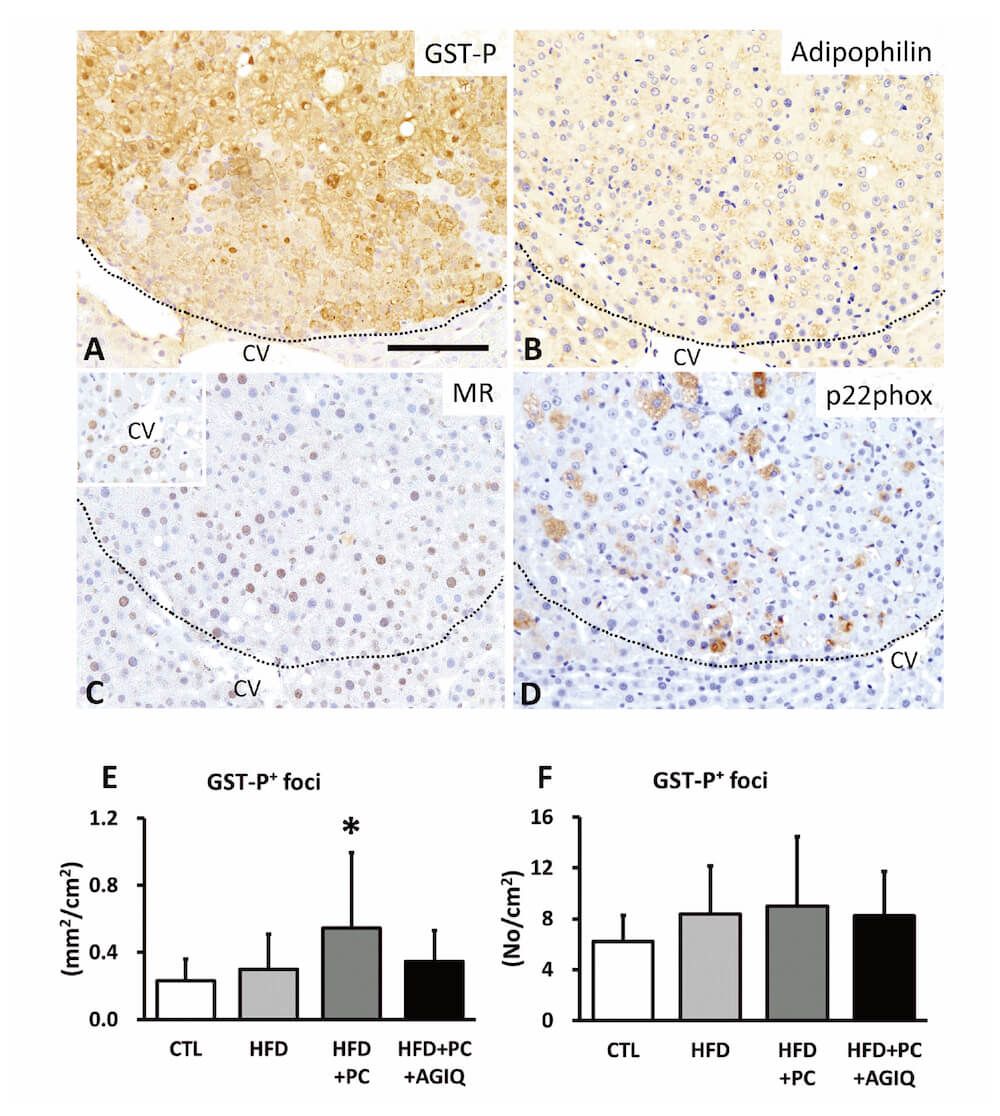
Fig. 4
Immunohistochemistry and quantitative analysis of preneoplastic foci in the liver of rats fed with basal diet (CTL) or high fat diet (HFD) co-administered with potassium canrenoate (HFD+PC) and/or an antioxidant alpha-glycosyl isoquercitrin (HFD+PC+AGIQ). Immunohistochemistry of glutathione S-transferase placental (GST-P) (A), adipophilin (B), mineralocorticoid receptor (MR) (C), and p22phox (D). (C) Inset shows nuclear localization of MR in the centrilobular region. The area (E) and number (F) of GST-P+ foci per unit are shown. CV: Central vein. Columns represent mean and standard deviation. * p < 0.05 vs. CTL (Tukey’s or Steel-Dwass multiple comparison test). Bar = 100 μm
| Antigen | Distribution | CTL | HFD | HFD+PC | HFD+PC+AGIQ | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ki-67 | Inside foci | 2.2 | ± | 1.7 | 2.0 | ± | 1.6 | 1.8 | ± | 1.3 | 1.6 | ± | 1.2 |
| Outside foci | 8.0 | ± | 2.4 | 9.6 | ± | 3.4 | 7.3 | ± | 4.6 | 7.1 | ± | 3.0 | |
| Active caspase-3 | Inside foci | 0.0 | ± | 0.1 | 0.1 | ± | 0.1 | 0.1 | ± | 0.2 | 0.0 | ± | 0.1 |
| Outside foci | 0.4 | ± | 0.2 | 0.4 | ± | 0.2 | 0.4 | ± | 0.2 | 0.5 | ± | 0.2 | |
Abbreviations: AGIQ, alpha-glycosyl isoquercitrin; CTL, control; DEN, N-diethylnitrosamine; HFD, high fat diet; PC, potassium canrenoate. §: All animals were subjected to two-thirds partial hepatectomy at week 1 after starting PC treatment with or without AGIQ. Data are shown as the mean ± standard deviation (%).
DISCUSSION
MR activation by aldosterone or corticosterone is involved in several physiological processes, and inappropriate activation of the MR by environmental or genetic factors can trigger deleterious responses in various tissues via NOX activation, which ultimately leads to the development of hypertension, cardiovascular injury, obesity, and insulin resistance (Zennaro et al., 2009). These risk factors might play a role in the development of NAFLD/NASH (Cömert et al., 2001; Marchesini et al., 2003); however, the role of MR/NOX signaling is not fully understood in the liver diseases. The present study showed the effects of a MR antagonist PC on obesity, insulin resistance, renal damage (renal basophilic tubules), and liver lesions (steatosis and preneoplastic liver foci) in a steatosis-related early hepatocarcinogenesis model.
Insulin resistance is the pathophysiological hallmark of NAFLD (Cömert et al., 2001; Marchesini et al., 2003), and increased free fatty acid flux from adipose tissue to non-adipose organs leads to triglyceride accumulation and contributes to impaired glucose metabolism and insulin sensitivity in the non-adipose organs, muscle, and liver (Bugianesi et al., 2005). We showed insulin resistance in HFD-fed rats using a glucose tolerance test. The results were consistent with evidence that HFD reproduced insulin resistance in rats (Zang et al., 2015; Catalogna et al., 2016). Data suggest that excessive circulating aldosterone promotes the development of impaired insulin metabolic signaling (Whaley-Connell et al., 2010). We had hypothesized that PC might improve insulin resistance and obesity, but the beneficial effects of PC were demonstrated only in obesity under the present study condition.
Obesity results in an increased risk of developing chronic kidney diseases, including glomerular damage, renal inflammation, and fibrosis (Nagai et al., 2005; Serra et al., 2008). The renal basophilic changes induced by the HFD can represent tubular regeneration after cell damage or indicate early tubular atrophy or persistent injury (Frazier et al., 2012). The basophilic changes have not been reported in the HFD-fed rats to our knowledge; however, necrosis of proximal tubular cells was reported in rats when fed hypercaloric high-fat-high-fructose diet for 10 weeks (Nasriet al., 2015). We showed that PC had a potential of preventing HFD-induced basophilic tubules as well as obesity. The preventive effects of PC on the HFD-induced basophilic tubules may be related to MR/NOX signaling pathway (Whaley-Connell et al., 2010), since the basophilic tubules expressed MR and p22phox as demonstrated by immunohistochemistry. The immuno-positive reaction of MR in the cytoplasms and nuclei was confirmed in the distal tubules and collecting tubules in kidney sections, as reported previously (Lombès et al., 1990), and supporting the role of MR in sodium absorption and potassium retention by regulating Na-Cl cotransporter in these tubules (Shibata, 2017). The biological significance of nuclear localization of MR in basophilic tubules remains uncertain.
Wada et al. (2010) reported that SR was able to suppress hyperlipidemia and hepatic steatosis in mice fed a HFD with fructose-supplemented water by reducing the transcript levels of pro-inflammatory cytokines and lipogenic enzymes in liver tissues. In this study, a ballooning change of hepatocytes in NAS significantly decreased with the combined treatment with PC and the antioxidant AGIQ. In altered lipid metabolism genes (Pparg, Acox1, and Pnpla2) analysis using RT-PCR, Pnpla2 expression significantly increased with HFD with PC or in combination with AGIQ treatment. A transcript of Pnpla2 mediates hydrolysis of lipid droplet and reduces steatosis in the liver (Sanchez-Lazo et al., 2014). A decrease of ballooning change with PC in combination with AGIQ treatment may be related to a decrease in Pnpla2 expression and an increase of antioxidant Catalase gene levels, which reduce steatosis-related oxidative stress in hepatocytes. This interpretation was supported by a previous finding that antioxidants reduced ballooning change of hepatocytes induced by hepatotoxicant in rats through the improvement of cell membrane rigidity (Aranda et al., 2010).
The most unexpected finding was that PC increased the area of preneoplastic liver foci in a steatosis-related hepatocarcinogenesis model. Because of the expression of MR and p22phox in the liver foci, the effect of PC may be related to MR/NOX signaling pathway; however, the drug increased gene expression of NADPH oxidase subunits (p22phox, Polidip2, and NOX4) in hepatic samples. Therefore, PC may have a potential as tumor promoter in association with MR/NOX-mediated oxidative stress, even though the drug is known to be a MR antagonist (Nasriet al., 2015). This interpretation was supported by the finding that the antioxidant AGIQ has potential of preventing PC-increased liver foci, and the effects might be associated with an increased gene expression of antioxidant Catalase.
The signaling by MR is complicated, since there are two systems, genomic and non-genomic events for controlling cellular homeostasis by aldosterone and MR (Ong and Young, 2017). MR is localized in the cytoplasm in the absence of stimuli of aldosterone. Once activated, MR translocates to the nucleus and transcribes target genes, including the MR gene (Nr3c2). We did not demonstrate that PC significantly altered expression level of Nr3c2; however, PC in combination with AGIQ significantly increased the level as compared with the control. MR antagonized by PC might be rescued by PC and AGIQ cotreatment; however, the biological significance is still unknown. Alternatively, aldosterone and MR act via second messengers such as MAPKs, PI3K/AKT, and PKC/PKD, often by activating unrelated receptors in the absence of their ligands (Ong and Young, 2017). MR is widely distributed in systemic organs, including kidney, gastrointestinal tracts, adrenals, immune system, brain, bone, adipose tissue, heart, skeletal muscle, and skin (Odermatt and Kratschmar, 2012). Genomic and nongenomic signals by MR are well documented in the kidney and vessels, but not in the other organs, including the liver. Previous reports also suggested genomic and nongenomic contributions to generation of NOX (Ong and Young, 2017). Further studies on roles of MR by genomic and nongenomic means in HFD-mediated renal and hepatic lesions are required.
We cannot rule out the possibility of non-MR/NOX signaling-mediated effects on the liver promotion by PC. Progestins (progestogens) are classified as possibly carcinogenic to humans by the International Agency for Research on Cancer (IARC, 1999). Because of the general negative genotoxicity of these chemicals, they have been considered as non-genotoxic chemicals acting as tumor promoters (Schuppler and Günzel, 1979). A synthetic drug of this family, cyproterone acetate, is activated to a reactive species by the liver and forms DNA adducts and elicits DNA repair in hepatocytes from both rats and humans (Brambilla and Martelli, 2002). PC and cyproterone acetate share the 17-hydroxy-3-oxopregna-4,6-diene structure (Brambilla and Martelli, 2002). PC was found to induce DNA fragmentation, as detected by the Comet assay and DNA repair synthesis in primary cultures of hepatocytes obtained from rats and humans, but the effects were weaker in human hepatocytes than those in rat hepatocytes (Martelli et al., 1999). An (non-significant) increase of active-caspase-3-positive cells inside foci may be related to DNA damage by PC treatment in this study. However, the genotoxicity of PC is controversial at present. Any evidence of DNA repair and micronuclei formation was absent in the liver of rats treated with 1/2 LD50 of PC (Martelli et al., 2002). The authors also demonstrated that 100 mg/kg PC did not increase preneoplastic liver foci in rats initiated with 2-acetylaminofluorene and CCl4 (Martelli et al., 2002). The discrepancy on the effects of liver promotion was probably dependent on the study condition and animal models.
Under the present study condition, HFD induced obesity and insulin resistance, and also increased renal and hepatic damage which might be mediated by MR/NOX signaling. PC reduced renal damage probably acting as an MR antagonist but unexpectedly increased preneoplastic liver foci through other mechanism(s). These unexpected hepatic effects may be caused by induction of NOX following inappropriate inhibition of MR or DNA damage by PC. Further studies are required to resolve these differential effects of MR antagonism in each target tissue and to understand how MR/NOX signaling is involved in the development of steatotic liver and preneoplastic liver lesions in association with systemic obesity and insulin resistance.
ACKNOWLEDGMENT
The authors thank Mrs. Shigeko Suzuki for her technical assistance in preparing the histological specimens. This work was supported by the Japan Food Chemical Research Foundation.
Conflict of interest
The authors declare that there is no conflict of interest.
REFERENCES
- Angeli, P., Fasolato, S., Mazza, E., Okolicsanyi, L., Maresio, G., Velo, E., Galioto, A., Salinas, F., D’Aquino, M., Sticca, A. and Gatta, A. (2010): Combined versus sequential diuretic treatment of ascites in non-azotaemic patients with cirrhosis: results of an open randomised clinical trial. Gut, 59, 98-104.
- Aranda, M., Albendea, C.D., Lostalé, F., López-Pingarrón, L., Fuentes-Broto, L., Martínez-Ballarín, E., Reiter, R.J., Pérez-Castejón, M.C. and García, J.J. (2010): In vivo hepatic oxidative stress because of carbon tetrachloride toxicity: protection by melatonin and pinoline. J. Pineal Res., 49, 78-85.
- Bolondi, L., Piscaglia, F., Gatta, A., Salerno, F., Bernardi, M., Ascione, A., Ferraù, O., Sacerdoti, D., Visentin, S., Trevisani, F., Mazzanti, R., Donati, G., Arena, U., Gentilini, P. and Group, D.O.; DOC (Decrease Of Complications in cirrhosis) Study Group. (2006): Effect of potassium canrenoate, an anti-aldosterone agent, on incidence of ascites and variceal progression in cirrhosis. Clin. Gastroenterol. Hepatol., 4, 1395-1402.
- Bos, R., Mougenot, N., Médiani, O., Vanhoutte, P.M. and Lechat, P. (2004): Potassium canrenoate, an aldosterone receptor antagonist, reduces isoprenaline-induced cardiac fibrosis in the rat. J. Pharmacol. Exp. Ther., 309, 1160-1166.
- Brambilla, G. and Martelli, A. (2002): Are some progestins genotoxic liver carcinogens? Mutat. Res., 512, 155-163.
- Bugianesi, E., McCullough, A.J. and Marchesini, G. (2005): Insulin resistance: a metabolic pathway to chronic liver disease. Hepatology, 42, 987-1000.
- Catalogna, M., Fishman, S., Halpern, Z., Ben-Shlomo, S., Nevo, U. and Ben-Jacob, E. (2016): Regulation of glucose dynamics by noninvasive peripheral electrical stimulation in normal and insulin-resistant rats. Metabolism, 65, 863-873.
- Chatterjee, S., Ganini, D., Tokar, E.J., Kumar, A., Das, S., Corbett, J., Kadiiska, M.B., Waalkes, M.P., Diehl, A.M. and Mason, R.P. (2013): Leptin is key to peroxynitrite-mediated oxidative stress and Kupffer cell activation in experimental non-alcoholic steatohepatitis. J. Hepatol., 58, 778-784.
- Cömert, B., Mas, M.R., Erdem, H., Dinc, A., Saglamkaya, U., Cigerim, M., Kuzhan, O., Unal, T. and Kocabalkan, F. (2001): Insulin resistance in non-alcoholic steatohepatitis. Dig. Liver Dis., 33, 353-358.
- Di Meo, S., Reed, T.T., Venditti, P. and Victor, V.M. (2016): Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev., 2016, 1245049.
- Dongiovanni, P., Romeo, S. and Valenti, L. (2014): Hepatocellular carcinoma in nonalcoholic fatty liver: role of environmental and genetic factors. World J. Gastroenterol., 20, 12945-12955.
- Frazier, K.S., Seely, J.C., Hard, G.C., Betton, G., Burnett, R., Nakatsuji, S., Nishikawa, A., Durchfeld-Meyer, B. and Bube, A. (2012): Proliferative and nonproliferative lesions of the rat and mouse urinary system. Toxicol. Pathol., 40(Suppl), 14S-86S.
- Hara, S., Morita, R., Ogawa, T., Segawa, R., Takimoto, N., Suzuki, K., Hamadate, N., Hayashi, S.M., Odachi, A., Ogiwara, I., Shibusawa, S., Yoshida, T. and Shibutani, M. (2014): Tumor suppression effects of bilberry extracts and enzymatically modified isoquercitrin in early preneoplastic liver cell lesions induced by piperonyl butoxide promotion in a two-stage rat hepatocarcinogenesis model. Exp. Toxicol. Pathol., 66, 225-234.
- International Agency for Research on Cancer (IARC). (1999): Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 72, Hormonal Contraception and Post-Menopausal Hormonal Therapy, IARC, Lyon, France.
- Ito, N., Tamano, S. and Shirai, T. (2003): A medium-term rat liver bioassay for rapid in vivo detection of carcinogenic potential of chemicals. Cancer Sci., 94, 3-8.
- Jonassen, T.E., Petersen, J.S., Sørensen, A.M., Andreasen, F. and Christensen, S. (1998): Aldosterone receptor blockade inhibits increased furosemide-sensitive sodium reabsorption in rats with liver cirrhosis. J. Pharmacol. Exp. Ther., 287, 931-936.
- Kleiner, D.E., Brunt, E.M., Van Natta, M., Behling, C., Contos, M.J., Cummings, O.W., Ferrell, L.D., Liu, Y.C., Torbenson, M.S., Unalp-Arida, A., Yeh, M., McCullough, A.J. and Sanyal, A.J.; Nonalcoholic Steatohepatitis Clinical Research Network. (2005): Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology, 41, 1313-1321.
- Kono, H., Rusyn, I., Yin, M., Gäbele, E., Yamashina, S., Dikalova, A., Kadiiska, M.B., Connor, H.D., Mason, R.P., Segal, B.H., Bradford, B.U., Holland, S.M. and Thurman, R.G. (2000): NADPH oxidase-derived free radicals are key oxidants in alcohol-induced liver disease. J. Clin. Invest., 106, 867-872.
- Liu, W., Baker, R.D., Bhatia, T., Zhu, L. and Baker, S.S. (2016): Pathogenesis of nonalcoholic steatohepatitis. Cell. Mol. Life Sci., 73, 1969-1987.
- Livak, K.J. and Schmittgen, T.D. (2001): Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Method. Methods, 25, 402-408.
- Lombès, M., Farman, N., Oblin, M.E., Baulieu, E.E., Bonvalet, J.P., Erlanger, B.F. and Gasc, J.M. (1990): Immunohistochemical localization of renal mineralocorticoid receptor by using an anti-idiotypic antibody that is an internal image of aldosterone. Proc. Natl. Acad. Sci. USA, 87, 1086-1088.
- Marchesini, G., Bugianesi, E., Forlani, G., Cerrelli, F., Lenzi, M., Manini, R., Natale, S., Vanni, E., Villanova, N., Melchionda, N. and Rizzetto, M. (2003): Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology, 37, 917-923.
- Martelli, A., Carrozzino, R., Mattioli, F., Bucci, G., Lamarino, G. and Brambilla, G. (2002): DNA damage in tissues of rat treated with potassium canrenoate. Toxicology, 171, 95-103.
- Martelli, A., Mattioli, F., Carrozzino, R., Ferraris, E., Marchese, M., Angiola, M. and Brambilla, G. (1999): Genotoxicity testing of potassium canrenoate in cultured rat and human cells. Mutagenesis, 14, 463-472.
- Nagai, J., Christensen, E.I., Morris, S.M., Willnow, T.E., Cooper, J.A. and Nielsen, R. (2005): Mutually dependent localization of megalin and Dab2 in the renal proximal tubule. Am. J. Physiol. Renal Physiol., 289, F569-F576.
- Nasri, R., Abdelhedi, O., Jemil, I., Daoued, I., Hamden, K., Kallel, C., Elfeki, A., Lamri-Senhadji, M., Boualga, A., Nasri, M. and Karra-Châabouni, M. (2015): Ameliorating effects of goby fish protein hydrolysates on high-fat-high-fructose diet-induced hyperglycemia, oxidative stress and deterioration of kidney function in rats. Chem. Biol. Interact., 242, 71-80.
- Nie, H., Li, J., Yang, X.M., Cao, Q.Z., Feng, M.X., Xue, F., Wei, L., Qin, W., Gu, J., Xia, Q. and Zhang, Z.G. (2015): Mineralocorticoid receptor suppresses cancer progression and the Warburg effect by modulating the miR-338-3p-PKLR axis in hepatocellular carcinoma. Hepatology, 62, 1145-1159.
- Nyska, A., Hayashi, S.M., Koyanagi, M., Davis, J.P., Jokinen, M.P., Ramot, Y. and Maronpot, R.R. (2016): Ninety-day toxicity and single-dose toxicokinetics study of alpha-glycosyl isoquercitrin in Sprague-Dawley rats. Food Chem. Toxicol., 97, 354-366.
- Odermatt, A. and Kratschmar, D.V. (2012): Tissue-specific modulation of mineralocorticoid receptor function by 11β-hydroxysteroid dehydrogenases: an overview. Mol. Cell. Endocrinol., 350, 168-186.
- Ong, G.S. and Young, M.J. (2017): Mineralocorticoid regulation of cell function: the role of rapid signalling and gene transcription pathways. J. Mol. Endocrinol., 58, R33-R57.
- Ozaki, K., Mahler, J.F., Haseman, J.K., Moomaw, C.R., Nicolette, M.L. and Nyska, A. (2001): Unique renal tubule changes induced in rats and mice by the peroxisome proliferators 2,4-dichlorophenoxyacetic acid (2,4-D) and WY-14643. Toxicol. Pathol., 29, 440-450.
- Sanchez-Lazo, L., Brisard, D., Elis, S., Maillard, V., Uzbekov, R., Labas, V., Desmarchais, A., Papillier, P., Monget, P. and Uzbekova, S. (2014): Fatty acid synthesis and oxidation in cumulus cells support oocyte maturation in bovine. Mol. Endocrinol., 28, 1502-1521.
- Schuppler, J. and Günzel, P. (1979): Liver tumors and steroid hormones in rats and mice. Arch. Toxicol. Suppl., 2, 181-195.
- Serra, A., Romero, R., Lopez, D., Navarro, M., Esteve, A., Perez, N., Alastrue, A. and Ariza, A. (2008): Renal injury in the extremely obese patients with normal renal function. Kidney Int., 73, 947-955.
- Shibata, S. (2017): 30 YEARS OF THE MINERALOCORTICOID RECEPTOR: mineralocorticoid receptor and NaCl transport mechanisms in the renal distal nephron. J. Endocrinol., 234, T35-T47.
- Thiesson, H.C., Jensen, B.L., Bistrup, C., Ottosen, P.D., McNeilly, A.D., Andrew, R., Seckl, J. and Skøtt, O. (2007): Renal sodium retention in cirrhotic rats depends on glucocorticoid-mediated activation of mineralocorticoid receptor due to decreased renal 11beta-HSD-2 activity. Am. J. Physiol. Regul. Integr. Comp. Physiol., 292, R625-R636.
- Wada, T., Kenmochi, H., Miyashita, Y., Sasaki, M., Ojima, M., Sasahara, M., Koya, D., Tsuneki, H. and Sasaoka, T. (2010): Spironolactone improves glucose and lipid metabolism by ameliorating hepatic steatosis and inflammation and suppressing enhanced gluconeogenesis induced by high-fat and high-fructose diet. Endocrinology, 151, 2040-2049.
- Whaley-Connell, A., Johnson, M.S. and Sowers, J.R. (2010): Aldosterone: role in the cardiometabolic syndrome and resistant hypertension. Prog. Cardiovasc. Dis., 52, 401-409.
- Yoshida, T., Murayama, H., Kawashima, M., Nagahara, R., Kangawa, Y., Mizukami, S., Kimura, M., Abe, H., Hayashi, S.M. and Shibutani, M. (2017): Apocynin and enzymatically modified isoquercitrin suppress the expression of a NADPH oxidase subunit p22phox in steatosis-related preneoplastic liver foci of rats. Exp. Toxicol. Pathol., 69, 9-16.
- Zang, S.S., Song, A., Liu, Y.X., Wang, C., Song, G.Y., Li, X.L., Zhu, Y.J., Yu, X., Li, L., Liu, C.X., Kang, J.C. and Ren, L.P. (2015): Chinese medicine Jinlida (JLD) ameliorates high-fat-diet induced insulin resistance in rats by reducing lipid accumulation in skeletal muscle. Int. J. Clin. Exp. Med., 8, 4620-4634.
- Zennaro, M.C., Caprio, M. and Fève, B. (2009): Mineralocorticoid receptors in the metabolic syndrome. Trends Endocrinol. Metab., 20, 444-451.