Gum ghatti, a polysaccharide of natural origin, is used in foods as a thickening, gelling, emulsifying and stabilizing agent. In a 90-day toxicity study following Organization for Economic Co-operation and Development (OECD) Guideline #408, male and female Sprague–Dawley rats were exposed to 0 (control), 0.5, 1.5 and 5% gum ghatti in AIN-93M basal diet. Expected changes included increased full and empty cecal weights in 5% groups. Incidentally 2/10 females from the 5% gum ghatti group had a single colon ulcer with associated acute inflammation. In a second 90-day study increased cecal weights were present in Sprague–Dawley females exposed to 5% gum ghatti in AIN-93M and NIH-07 basal diets. A single colon ulcer with associated acute inflammation occurred in 1/20 control females given AIN-93M basal diet. The colon ulcers were considered a sporadic change possibly attributable to AIN-93M basal diet. In the second study a few statistically significant alterations in clinical chemistry were considered sporadic and unrelated to treatment. Feed consumption among treated and control groups was similar for each sex. Gum ghatti intake at the 5% dietary level ranged from 3044 to 3825 mg/kg body weight/day. The 5% dietary administration was a NOAEL in both studies. NOAELs for males and females in the first study were 3044 and 3309 mg/kg/day, respectively. NOAELs for females in the second study were 3670 and 3825 mg/kg/day for AIN-93M and NIH-07 diets, respectively.
Keywords
Gum, Food additive, Cecal enlargement, AIN-93M diet
1. Introduction
Gum ghatti is one of several gums used commercially in foods as emulsifiers, thickeners, and stabilizers. Polysaccharide gums are a source of soluble fiber and are important additives to replace calorific foods. Gum ghatti is a complex polysaccharide obtained as an exudate of the Anogeissus latifolia tree, native to the dry deciduous forests of India (Sakai et al., 2012). It was originally considered as a substitute for gum arabic in the early 1900s but was not commercially developed because of batch-to-batch variability (Kaur et al., 2009). Gum ghatti naturally exists as mixed salts of calcium, magnesium, potassium, and sodium (Kaur et al., 2009). Hydrolysis produces L-arabinose, D-galactose, D-mannose, D-xylose, D-glucuronic acid (48:29:10:5:10 M ratios), and less than 1% L-rhamnose (Aspinall et al., 1965; Kaur et al., 2009; Sakai et al., 2012; Tischer et al., 2002). More recently a new gum ghatti product, Gatifolia, derived by a non-chemical physical procedure of spray-drying the dissolved, filtered, and sterilized starting material has been produced. This process yields a product with consistent batch-to-batch quality, superior rheological properties, acid resistance, and salt tolerance (Al-Assaf et al., 2008, 2009; Ido et al., 2008; Pszczola and Banasiak, 2006). The new gum ghatti product also has greater water solubility compared to many other polysaccharides (Kaur et al., 2009). Consequently, gum ghatti with its low viscosity has excellent emulsification properties at lower concentrations than gum arabic (Al-Assaf et al., 2008, 2009; Sakai et al., 2012) with potentially important applications in the food industry.
As a complex polysaccharide with an approximate molecular weight of 12,000 Da, gum ghatti is expected to escape degradation in the stomach and small intestine of humans and animals and undergo anaerobic microbial fermentation in the cecum and colon. The feeding of similar organic gums, including gum arabic, guar gum, rhamsan gum as well as other oligosaccharides, to laboratory rats results in increased cecal weight and size (Ali et al., 2009; Doi et al., 2006; Hagiwara et al., 2010; Levrat et al., 1991; Phillips, 1998; Tulung et al., 1987), increased blood flow to the cecum (Tulung et al., 1987; Younes et al., 1995), increased cecal bacterial proliferation (May et al., 1994; Walter et al., 1988), and bacterial fermentation to short chain volatile fatty acids with a cecal pH decrease to 6 (Levrat et al., 1991; McLean Ross et al., 1984; Walter et al., 1988). Complete cecal fermentation of gum arabic has been noted in humans (Phillips, 1998). Both gum arabic and gum ghatti are fermented in the human large intestine by Bacteriodes longum (Salyers et al., 1977).
Gum ghatti is marketed as a food additive in Japan without limitation (JMHLW, 2009) and was assigned GRAS status in the United States in 1965 by the Flavor and Extract Manufacturers Association (FEMA No. 2519; Hall and Oser, 1965) and in 1977 by the FDA (21CFR184.1333; FDA, 2010). Because of limited toxicity data on gum ghatti, the Joint FAO/WHO Expert Committee on Food Additives (JECFA) has not as yet set dietary intake limits. Utilizing an internationally adopted battery of tests to evaluate potential somatic and germ cell genetic risks to humans, gum ghatti was recently reported to have no evidence of genotoxic potential when given at the maximum OECD recommended guidelines (Hobbs et al., 2012).
The purpose of this report is to document the lack of systemic and organ specific toxicity of gum ghatti following administration to rats at up to 5% in the diet.
2. Materials and methods
2.1. Animal study and study design
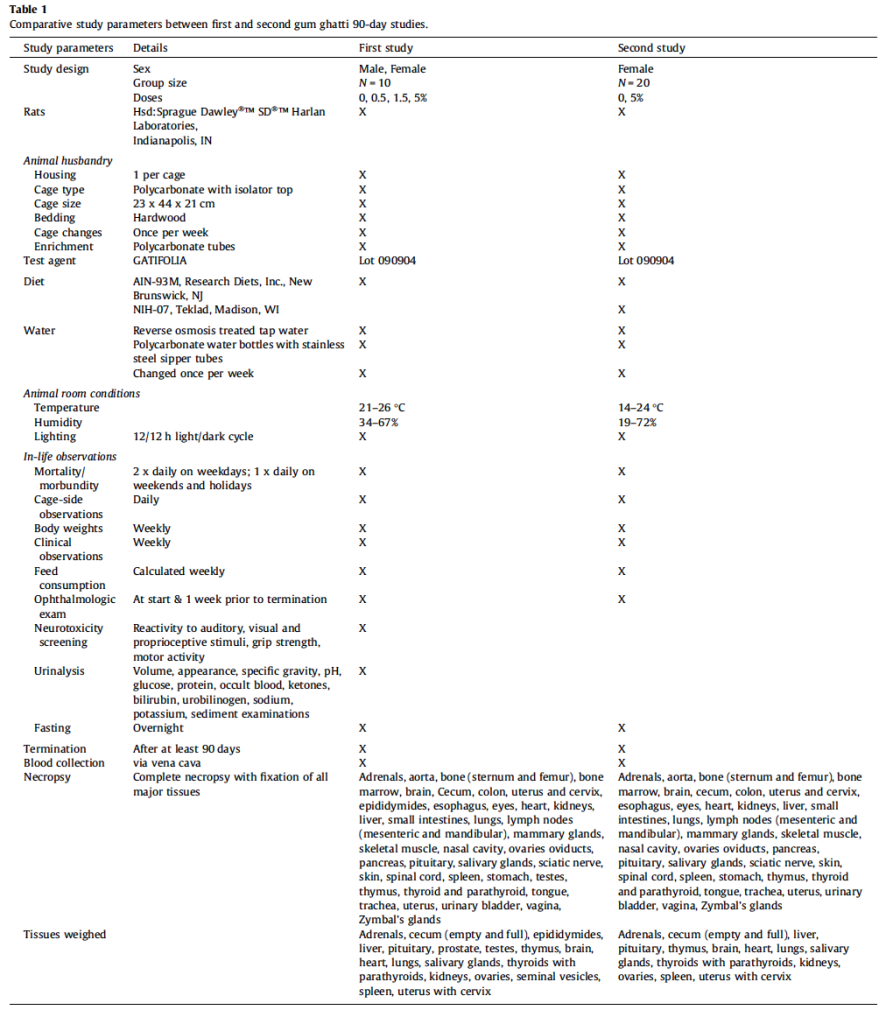
A standard 90-day Good Laboratory Practices (GLP) toxicity study was conducted in male and female Sprague Dawley rats according to OECD guideline #408 in which gum ghatti was prepared in AIN-93M diet. Based upon incidentally occurring colon ulcers in two females in the highest-dose (5% in diet), a second 90- day Good Laboratory Practices (GLP) toxicity study was conducted in females using the 5% high dose in two different diets, AIN-93M and NIH-07. Both studies were conducted within the same Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited specific pathogen free facility. All procedures were in compliance with Animal Welfare Act Regulations (9 CFR 1-4) and the Guide for the Care and Use of Laboratory Animals (ILAR, 2011) and with approval by the Integrated Laboratory Systems (ILS), Inc. Institutional Animal Care and Use Committee. In the first study groups of 10 males and 10 females were given gum ghatti at 0, 0.5, 1.5 and 5% in diet for at least 90 days. In the second study groups of 20 females were given gum ghatti in AIN-93M and NIH-07 at 0 and 5% for at least 90 days. Study parameters were essentially identical with minor exceptions (Table 1). Rats ordered for the second study were specifically requested to be non-littermates.
2.2. Test agent
Gum ghatti (Gatti Gum SD, previously called GATIFOLIA SD) was provided by San-Ei Gen, F.F.I., Inc. (Osaka, Japan), as a gray to reddish-gray powder and mixed in diet at Research Diets, Inc. (New Brunswick, NJ). For the first study, two batches of dosed diet were prepared taking into account a 58-day stability in feed. Similarly, two batches of dosed diet were prepared for use in the second 90-day study. The same lot of gum ghatti was used in both studies. Dose formulation analyses were conducted by OpAns, LLC (Durham, NC) and were within acceptable ranges in both studies.
2.3. Statistical analysis
Group mean, and standard deviations were calculated using Microsoft Excel. All data were analyzed (final body weight, body weight gain, feed consumption
3. Results
3.1. First study
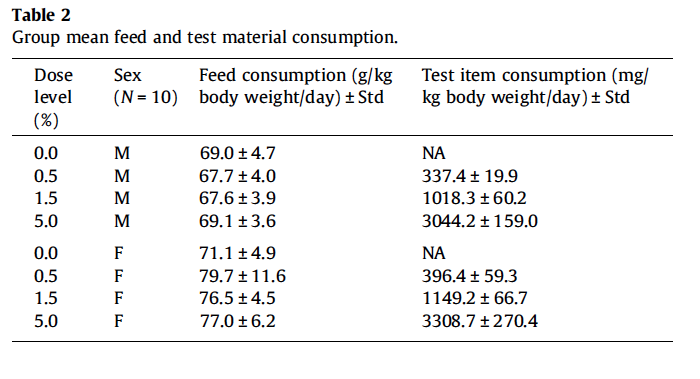
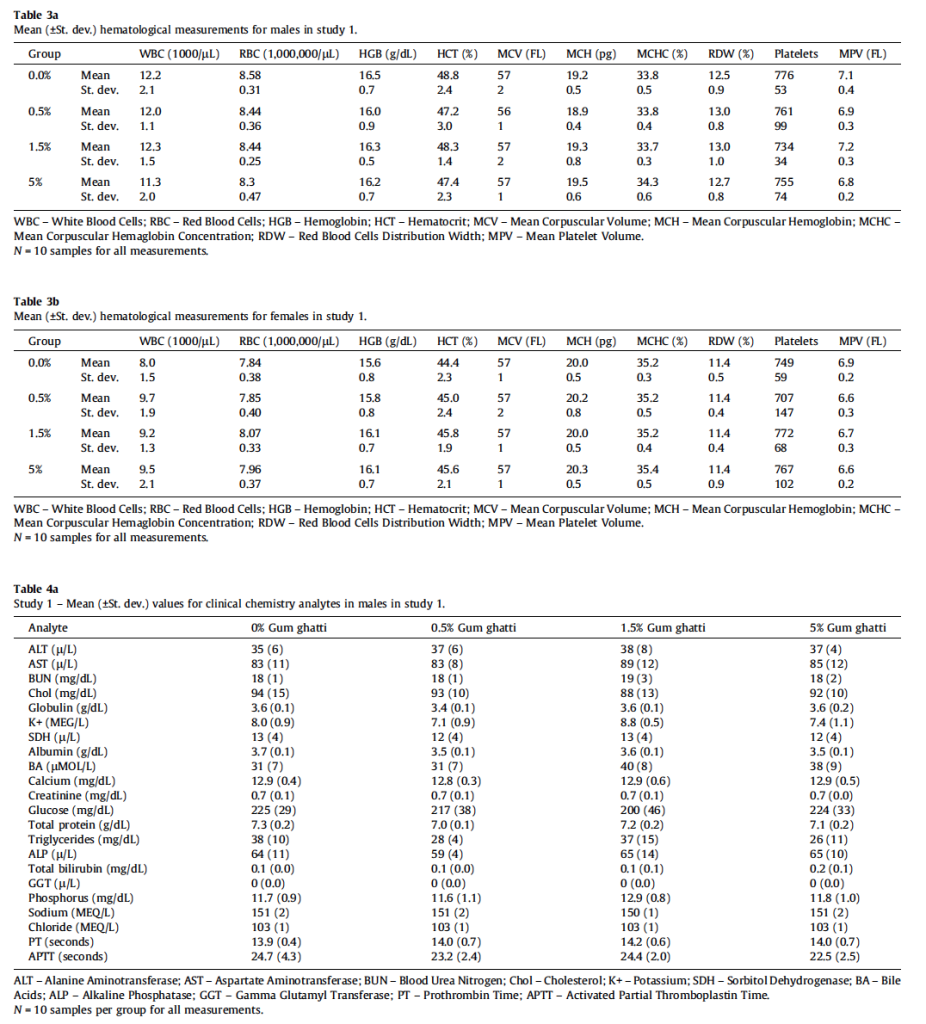
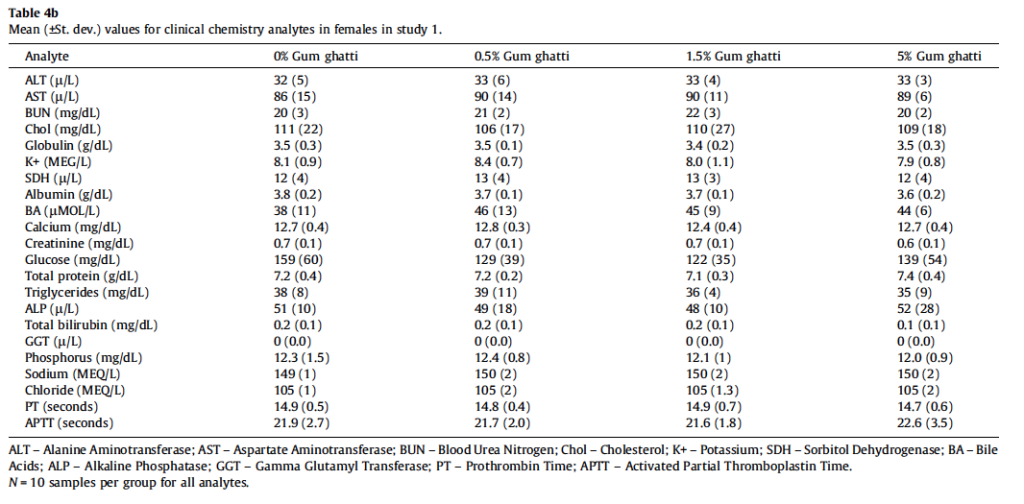
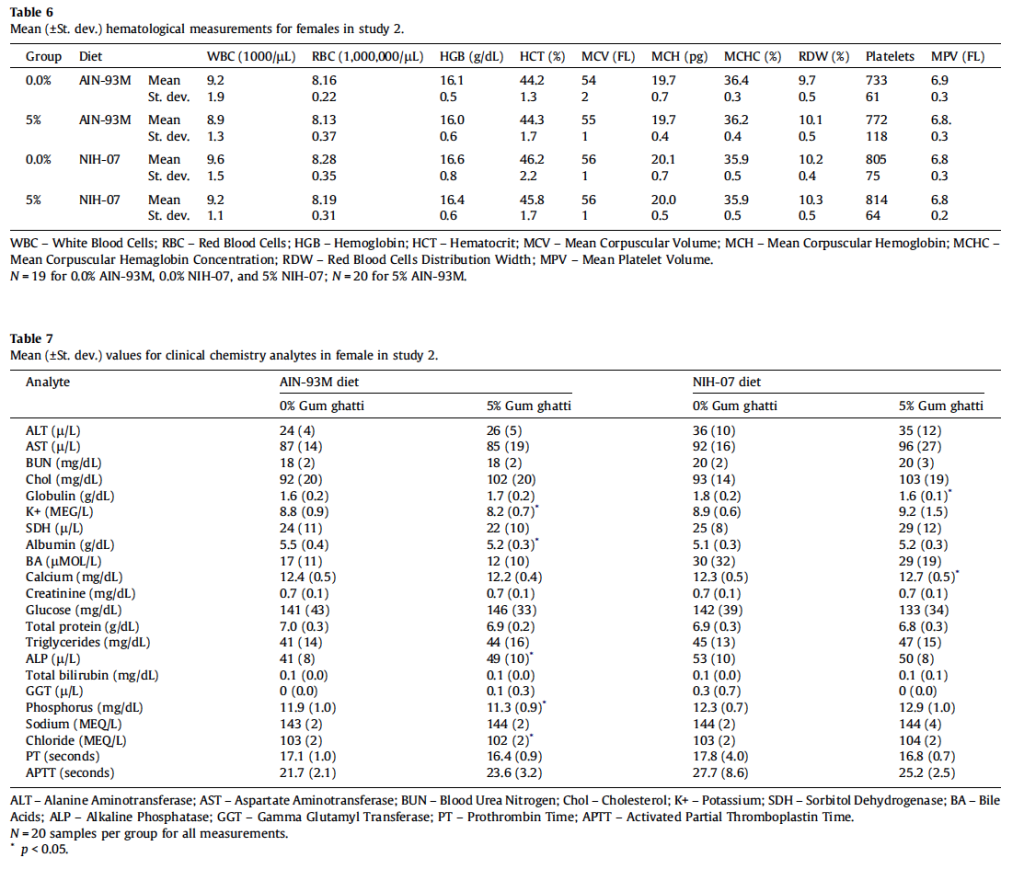
All animals survived until scheduled euthanasia with no clinical signs of toxicity in any animal. The majority of measured parameters did not differ significantly between gum ghatti-exposed and control rats. Mean feed consumption (Fig. 1A and Table 2), body weight (Fig. 1B), body weight gains and tissue weights (adrenal, brain, epididymides, heart, kidney, liver, lung, ovaries, pituitary, prostate, salivary glands, seminal vesicles, spleen, testes, thymus, thyroid/parathyroid and uterus/cervix) were not affected by treatment. Mean gum ghatti consumption is presented in Table 2. Mean hematological measurements and clinical chemistry analytes are presented in Tables 3a, 3b, 4a, and 4b. Ophthalmologic and neurological examination, urinalysis, plasma clotting times, histopathology of tissues from 40 sites and clinical chemistry endpoints were unaffected by gum ghatti. A statistically significant (p < 0.05) increase in monocytes was flagged in treated females due to a low control value but was within normal range for this measurement. No other hematological effects attributable to gum ghatti exposure were identified.
Exposure to high dose (5%) gum ghatti produced minimal to moderate changes in the cecum of male and colon of female rats. Cecal crypt hyperplasia was observed in 6/10 male rats but evaluation was problematic in that cecal mucosa orientation was not consistently perpendicular. No significant lesions were present in female cecum. The 5% dose of gum ghatti was also associated with significant increases in full and empty cecum weights (Fig. 1C and D). Focal ulceration with acute inflammation of the colon was present in 2/10 female rats exposed to the highest dose (Fig. 2); however no significant lesions were observed in male colon. A thorough re-examination of fixed colon wet tissues from all rats failed to identify any additional lesions. Because of suspicion that the focal colonic ulcerations may have been associated with the AIN-93M diet or the chance that intrinsically susceptible littermates were randomly assigned to the same group, a second study was undertaken to specifically focus on histopathology of female colon.
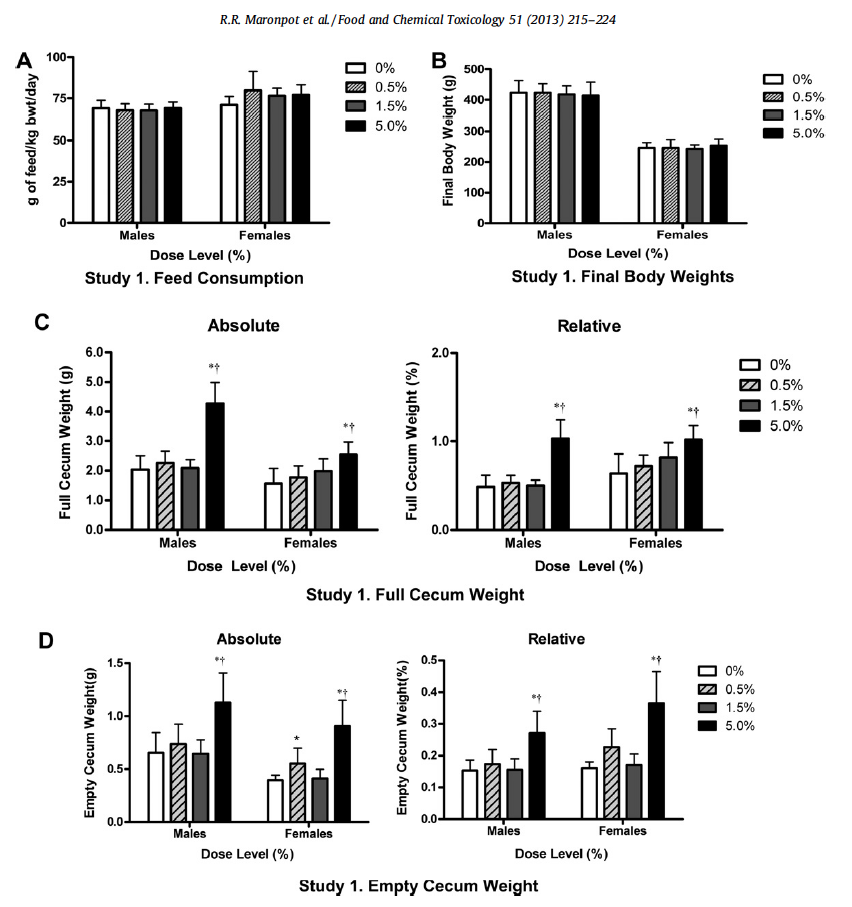
Fig. 1. First study feed consumption (A), final body weight (B), full (C) and empty (D) cecal weights from rats administered 0, 0.5, 1.5 or 5% gum ghatti in AIN-93M diet for at least 90 days. Bars represent mean ± SD of 10 animals/group. ⁄ Statistically significant increase compared to vehicle control group (Dunnett’s test, p < 0.05). Statistically significant dose dependent increase following exposure to gum ghatti (trend test analysis, p < 0.05).
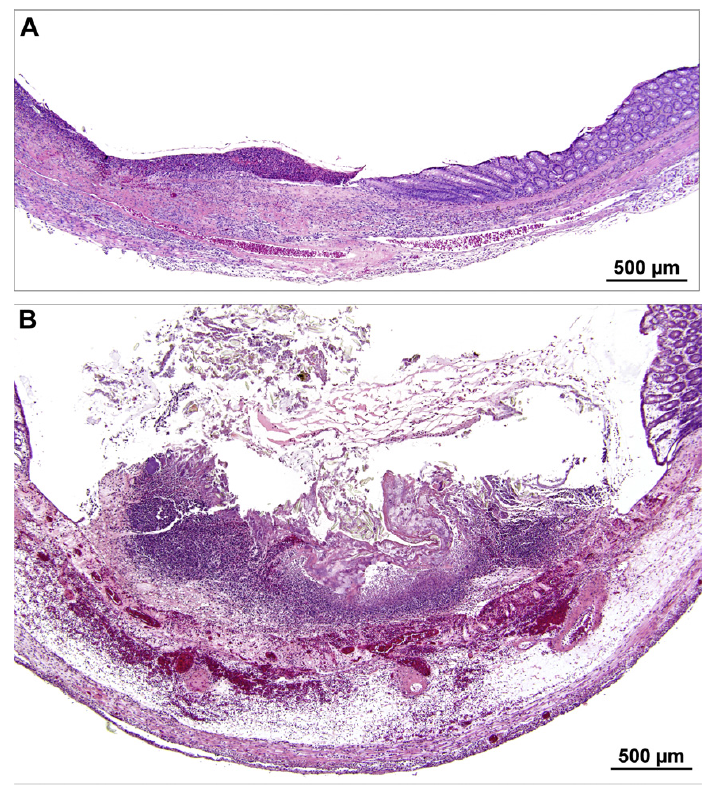
Fig. 2. (A and B) Photomicrographs of colon ulcers from 2 female rats administered 5% gum ghatti in AIN-93M diet for at least 90 days. Both ulcers are associated with submucosal inflammation. In both cases relatively normal mucosa is present on the right edge. Panel B is more acute with submucosal edema and hemorrhage and exfoliating necrotic surface mucosa.
3.2. Second study
The purpose of this study was to determine if exposure to 5% dietary gum ghatti for at least 90 days was causally associated with colon changes in female rats. Differences in study parameters from the first study included use of only female rats, exposure to only 0 and 5% gum ghatti, and use of two diets, viz., AIN-93M and NIH-07, with 20 rats per group. Neuropathology assessment and urinalysis were not carried out in the second study and histopathology assessment was restricted to cecum and colon. To maximize cecal and colonic mucosal tissue orientation, these tissues were opened and fixed flat on cardstock. Otherwise, study parameters were similar to those in the first study.
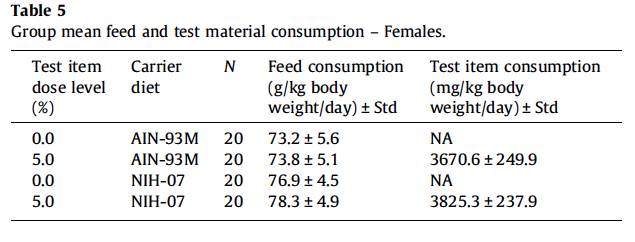
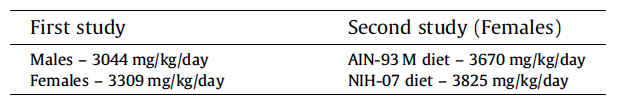
All animals survived until scheduled euthanasia with no clinical signs of toxicity in any animal. No gum ghatti-associated changes were observed in feed consumption (Fig. 3A and Table 5), final body weight (Fig. 3B), body weight gain, and ophthalmological parameters. Feed consumption was measured weekly and showed that the amount of test item consumed in the 2nd study (Table 5) was equivalent to the amount consumed in the 5% dose group of the 1st study (See Table 2). Mean gum ghatti consumption is presented in Table 5. Hematological measurements were similar between controls and treated rats and also similar between the two diets (Table 6). No differences in tissue weights relative to controls were observed for: adrenal, brain, heart, kidney, liver, lung, ovary, pituitary, salivary gland, spleen, thymus, thyroid with parathyroid and uterus with cervix. Sporadic statistical differences in clinical chemistry analytes with no correspondence between the first and second study or between the two diets in the second study were flagged (Table 7).
Full cecal weights (absolute and relative) were increased in rats exposed to gum ghatti in AIN-93M diet compared to carrier diet alone, but not in rats exposed to gum ghatti in a NIH-07 diet (Fig. 3C). Increased empty cecal weights (absolute and relative) were observed in gum ghatti (5.0%) exposed animals (both diets) compared to concurrent controls (Fig. 3D). The increased full and empty cecal weights in females exposed to gum ghatti in AIN- 93M diet were consistent with the findings in the first study. In the second study, histopathological analysis of hematoxylin and eosin-stained tissue sections showed no gum ghatti-associated lesions in the colon or cecum of animals fed either diet. One rat fed AIN-93M carrier diet alone had a mucosal ulcer with hemorrhage, mild submucosal edema and inflammation in the mid colon (Fig. 4).
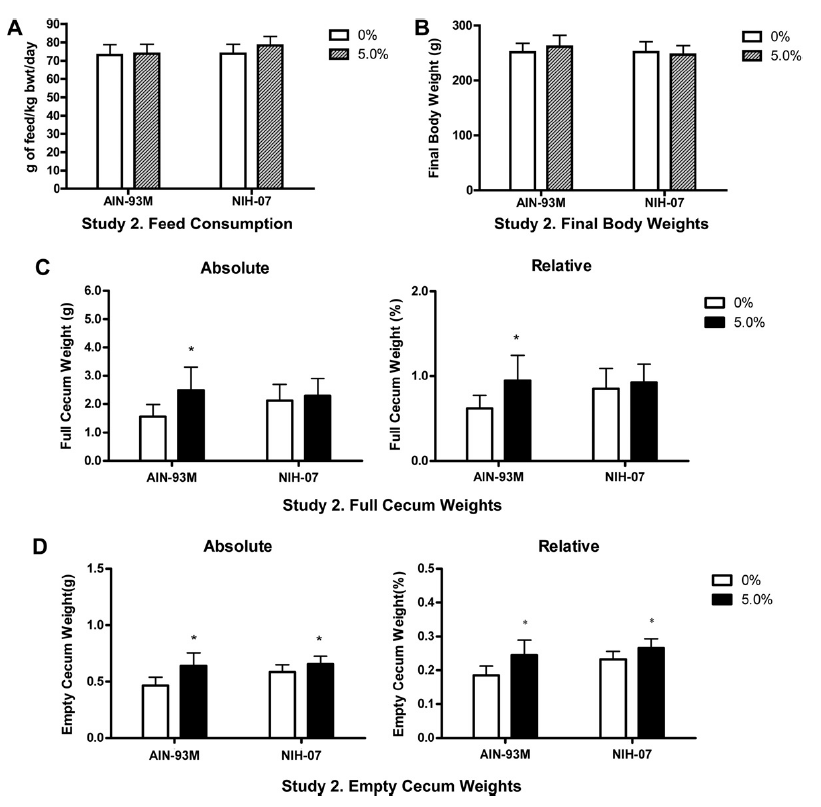
Fig. 3. Second study feed consumption (A), final body weight (B), full (C) and empty (D) cecal weights from female rats administered 0 or 5% gum ghatti in AIN-93M or NIH-07 diet for at least 90 days. Bars represent mean ± SD of 20 animals/group. ⁄ Statistically significant increase compared to vehicle control group (Dunnett’s test, p < 0.05).
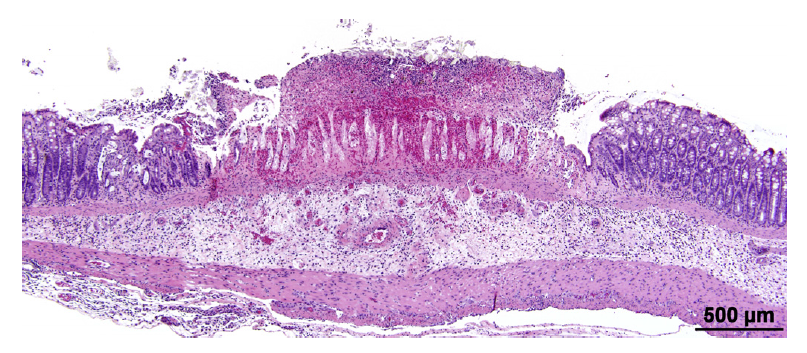
Fig. 4. A focal colonic ulcer in a control female rat administered AIN-93M basal diet for at least 90 days. Relatively normal mucosa is present on the right and left of the centrally located active ulcer. There is submucosal edema and inflammatory cell infiltration. The affected mucosa is covered by exfoliated necrotic mucosa overlying a band of mucosal hemorrhage.
3.3. Pathology Peer Review and Post-Study Pathology Working Group (PWG)
Pathology findings from both 90-day studies were independently peer reviewed and no additional findings were identified. At the conclusion of the second study, a panel of experts in rodent pathology formed a PWG to specifically evaluate the colon lesions and to assess the histologic evidence for changes in the cecal crypts noted in the first study. The three instances of focal colonic ulceration with associated acute inflammation were similar and represented an active response that occurred at the end of the studies.
The PWG concluded that the colon ulceration was a sporadic event not causally associated with gum ghatti and, based on re-examination of cecal tissue from both studies, could not confirm cecal crypt hyperplasia diagnosed in the 6/10 males in the first study. The PWG confirmed the presence of colon ulcers and concluded changes in cecal crypts in the first study were within normal variability.
4. Discussion
4.1. Genotoxicity and cecal enlargement
Based on genotoxicity testing (bacterial reverse mutation assays, chromosomal aberration assay, combined micronucleus/Comet assay in mice), there is no evidence of genotoxic potential of gum ghatti given up to maximum concentrations recommended by OECD guidelines (Hobbs et al., 2012). In two 90-day studies, administration of the same lot of gum ghatti was associated with an expected adaptive cecal enlargement but no toxicologically relevant changes. Cecal enlargement is a well-documented adaptive change associated with dietary administration of fermentable water-soluble complex polysaccharide fiber components present in a variety gums commonly used as food additives (McLean Ross et al., 1984;Tulung et al., 1987; Younes et al., 1995; Walter et al., 1988). One of these gums, gum arabic, produced cecal enlargement in humans (McLean Ross et al., 1983). The mechanism for the cecal enlargement has not been clearly defined but speculated to involve increased blood flow and increased bacterial content (May et al., 1994; Tulung et al., 1987; Walter et al., 1988; Younes et al., 1995).
4.2. First study
In the first 90-day study reported here, 2/10 female rats administered 5% gum ghatti had a single focal ulcer with associated acute inflammation in the colon. No other colon lesions were present in the females and no colon lesions were present in the males. Colon wet tissue from all male and female rats in this study were opened and thoroughly examined for additional lesions and none were found.
The histomorphologic features of the colon ulcers were consistent with acute lesions occurring toward the end of the 90-day study. A likely pathogenesis would start with focal mucosal damage, possibly secondary to alterations in bacterial flora known to be associated administration of gums (May et al., 1994; Walter et al., 1988). The inflammation reaction is consistent with acute bacterially induced mucosal damage. Based on unsubstantiated hearsay from a pathology colleague, we speculated that the occurrence of a single colonic ulcer in 2/10 high dose females might have been associated with the AIN-93M diet. A thorough review of published literature failed to identify any association of AIN diets with colonic ulcers in rats. Alternatively, the ulcers could be a consequence of inadvertently having two littermates with this predisposition in the same experimental group. We are not able to identify a reason for colonic ulcers occurring only in females. An occasional spontaneous adverse lesion is sometimes seen in typical 90-day toxicity studies and, if truly spontaneous, would be expected to occur more frequently in treated animals simply because there are typically 3–4 times more treated animals in a study than controls. However, the occurrence in 2/10 high dose rats of one sex created sufficient concern that we elected to perform a second 90-day study with focus on female rat colon. To address our speculation, we requested no littermates from the animal supply vendor, we increased the group size from 10 to 20 rats, and we included a second non-purified standard laboratory diet in addition to the purified AIN-93M diet used in the first study. The second study employed similar endpoints to the first study with minor exceptions (See Table 1).
4.3. Second study
In the second 90-day study, the cecum and colon were opened and fixed flat on an index card to permit thorough examination of the mucosa during necropsy and to insure perpendicular orientation of the mucosa for preparation of histologic sections. A focal colonic ulcer with associated acute inflammation was seen in 1/20 control rats receiving the AIN-93M diet. The histomorphology of the colon ulcer was similar to that seen in the first 90-day study. The expected cecal enlargement was present and somewhat more dramatic in the rats administered gum ghatti in AIN-93M than in the rats administered gum ghatti in NIH-07 diet. The few clinical chemistry endpoints associated with statistical flags (Table 7) are considered spurious and unrelated to administration of gum ghatti since they were not consistent between studies, sex, or diet; were not associated with alteration of biologically related endpoints; and were too small to be clinically or biologically relevant. Spuriously occurring endpoints with statistical flags, particularly common in high precision clinical pathology analyte measurements, occur in most toxicity studies and may represent false positive and/or non-adverse changes (Hamada et al., 1998; James, 1993; Lewis et al., 2002). Increasing the number of rats per group to 20 would decrease variability and thus increase the chance of generating statistical flags. Furthermore, interpretation of statistically significant endpoints as toxicologically relevant requires con- firmation with associated relevant clinical pathology endpoints, expected consequences of the test agent, and/or related histopathology (Lewis et al., 2002).
4.4. Pathology Working Group (PWG)
A PWG panel of experts in rodent toxicologic pathology was charged with identifying gum ghatti-related changes in the two 90-day studies and to offer an opinion relative to the potential toxicity of gum ghatti. The PWG review included examination of the colon lesions and 50% of the cecum and colon sections from both 90-day studies. The PWG confirmed the presence of the colonic ulcers with associated acute inflammation. The PWG panel concluded that cecal crypt elongation and hyperplasia reported in the first 90-day study were within the range of normal variability and may have been affected by variable handling of the tissues during fixation, trimming, embedding, and preparation of tissue sections. The PWG panel concluded that there was no evidence that the focal ulceration with acute inflammation observed in the colon was directly associated with the administration of gum ghatti. The PWG also did not consider the increase in cecal organ weight to be toxicologically relevant.
4.5. Potential pathogenesis of colon ulcers
Since the colonic ulcers with associated inflammation are acute lesions without attendant reparative hyperplasia, the initiating event would have occurred within one to three days prior to necropsy. We speculate that the effects of gum ghatti on the intestinal track altered the ecology of the gut microbiome resulting in changes in numbers, diversity and stability of commensal bacteria, a condition referred to as dysbiosis. Increased bacterial flora in rats has been associated with dietary administration of other gums (May et al., 1994; Walter et al., 1988). Dysbiosis is consistent with the observed effect of gum ghatti on cecal enlargement and can lead to development of inflammation (Yang and Pei, 2006). The initial change would likely involve bacterially induced mucosal damage leading to erosion and ulceration with the inflammatory response occurring secondary to the mucosal damage. It is further speculated that AIN-93M diet may facilitate intestinal dysbiosis more than NIH-07 diet.
5. Conclusions
Administration of gum ghatti to rats at up to 5% in the diet for at least 90-days caused an expected increase in cecal weight consistent with other reported dietary gum studies. No unequivocal morphological changes were associated with the enlarged ceca. The occurrence of focal colon lesions with associated acute inflammation in two females given 5% gum ghatti in AIN-93M diet (first study) and in one female control given AIN-93M basal diet (second study) is speculated but certainly not proven to be related to the AIN-93M diet. Colon ulceration was not present in males given 5% gum ghatti in AIN-93M diet in the first study or in females given gum ghatti in AIN-93M or NIH-07 diet in the second study. Based on the two peer reviewed studies and subsequent PWG evaluation of cecum and colon sections, the NOAELs for gum ghatti given at 5% in the diet are as follows:
Conflict of Interest
This work was conducted at ILS, Inc., and funded by San-Ei Gen, F. F. I., Inc., a manufacturer of gum ghatti. Maronpot Consulting LLC is a paid consultant for ILS, Inc. and San-Ei Gen, F.F.I., Inc.
Role of the Funding Source
ILS, Inc., was responsible for the study design, the collection, analysis, and interpretation of data and preparation of the final study report. The manuscript was prepared by R. Maronpot. The decision to submit the paper for publication was made by San-Ei Gen, F.F.I., Inc.
Acknowledgements
The authors thank the investigative toxicology staff, necropsy and histology teams of Integrated Laboratory Systems, Inc. for technical support and PWG members for their expertise and involvement in the Pathology Working Group. The use of animals in these studies complied with all relevant federal guidelines and were approved by an Institutional Animal Care and Use Committee.
References
Al-Assaf, S., Amar, V., Phillips, G., 2008. Characterization of gum ghatti and comparison with gum arabic. In: Williams, P.A., Phillips, G.O. (Eds.), Gums and Stabilisers for the Food Industry. Royal Society of Chemistry Publishing, Cambridge, pp. 280–290.
Al-Assaf, S., Phillips, G., Amar, V., 2009. Gum ghatti. In: Handbook of Hydrocolloids. CRC Press, Cambridge, pp. 477–494.
Ali, B.H., Ziada, A., Blunden, G., 2009. Biological effects of gum arabic: a review of some recent research. Food Chem. Toxicol. 47, 1–8.
Aspinall, G., Bhavanadan, V., Christensen, T., 1965. Gum ghatti (India gum). J. Chem. Soc. 2677, 2684.
Doi, Y., Ichihara, T., Hagiwara, A., Imai, N., Tamano, S., Orikoshi, H., Ogasawara, K., Sasaki, Y., Nakamura, M., Shirai, T., 2006. A ninety-day oral toxicity study of a new type of processed gum arabic, from Acacia tree (Acacia senegal) exudates, in F344 rats. Food Chem. Toxicol. 44, 560–566.
FDA, 2010. Code of Federal Regulations, Title 21, vol. 3, 21CFR184.1333.
Hagiwara, A., Imai, N., Doi, Y., Sano, M., Tamano, S., Omoto, T., Asai, I., Yasuhara, K., Hayashi, S.M., 2010. Ninety-day oral toxicity study of rhamsan gum, a natural food thickener produced from Sphingomonas ATCC 31961, in Crl:CD(SD)IGS rats. J. Toxicol. Sci. 35, 493–501.
Hall, R., Oser, B., 1965. Recent progress in the consideration of flavor ingredients under the Food Additives Amendment 3. GRAS Substances. Food Technol. 19, 151–197.
Hamada, C., Yoshino, K., Abe, I., Matsumoto, K., Nomura, M., Yoshimura, I., 1998. Detection of an outlier and evaluation of its influence in chronic toxicity studies. Drug Inf. J. 32, 201–212.
Hobbs, C.A., Swartz, C., Maronpot, R., Davis, J., Recio, L., Hayashi, S.M., 2012. Evaluation of the genotoxicity of the food additive, gum ghatti. Food Chem. Toxicol. 50, 854–860.
Ido, T., Ogasawara, T., Katayama, T., Sasaki, Y., Al-Assaf, S., Phillips, G.O., 2008. Emulsification properties of GATIFOLIA (gum ghatti) used for emulsions in food products. FFI J. 213, 365–370.
ILAR, 2011. Guide for the Care and Use of Laboratory Animals, eighth ed. National Academies Press, Washington, DC.
James, R., 1993. The relevance of clinical pathology to toxicology studies. Comp. Haematol. Int. 3, 190–195.
JMHLW, 2009. Japan’s Specifications and Standards for Food Additives, eighth ed. The Ministry of Health, Labour, and Welfare, Japan.
Kaur, L., Singh, J., Singh, H., 2009. Characterization of gum ghatti (Anogeissus latifolia): a structural and rheological approach. J. Food Sci. 74, E328–E332.
Levrat, M.A., Behr, S.R., Remesy, C., Demigne, C., 1991. Effects of soybean fiber on cecal digestion in rats previously adapted to a fiber-free diet. J. Nutr. 121, 672– 678.
Lewis, R.W., Billington, R., Debryune, E., Gamer, A., Lang, B., Carpanini, F., 2002. Recognition of adverse and nonadverse effects in toxicity studies. Toxicol. Pathol. 30, 66–74.
May, T., Mackie, R.I., Fahey Jr., G.C., Cremin, J.C., Garleb, K.A., 1994. Effect of fiber source on short-chain fatty acid production and on the growth and toxin production by Clostridium difficile. Scand. J. Gastroenterol. 29, 916–922.
McLean Ross, A.H., Eastwood, M.A., Brydon, W.G., Anderson, J.R., Anderson, D.M., 1983. A study of the effects of dietary gum arabic in humans. Am. J. Clin. Nutr. 37, 368–375.
McLean Ross, A.H., Eastwood, M.A., Brydon, W.G., Busuttil, A., McKay, L.F., 1984. A study of the effects of dietary gum arabic in the rat. Br. J. Nutr. 51, 47–56.
Phillips, G.O., 1998. Acacia gum (Gum Arabic): a nutritional fibre; metabolism and calorific value. Food Addit. Contam. 15, 251–264.
Pszczola, D., Banasiak, K., 2006. Enter IFT’s Magic Ingredient Kingdom. Institute of Food Technologists, pp. 45–92.
Sakai, E., Katayama, T., Ogasawara, T., Mizuno, M., 2012. Identification of Anogeissus latifolia Wallich and analysis of refined gum ghatti. J. Nat. Med.. http:// dx.doi.org/10.1007/s11418-012-0678-3.
Salyers, A.A., West, S.E., Vercellotti, J.R., Wilkins, T.D., 1977. Fermentation of mucins and plant polysaccharides by anaerobic bacteria from the human colon. Appl. Environ. Microbiol. 34, 529–533.
Tischer, C.A., Iacomini, M., Wagner, R., Gorin, P.A., 2002. New structural features of the polysaccharide from gum ghatti (Anogeissus latifola). Carbohydr. Res. 337, 2205–2210.
Tulung, B., Remesy, C., Demigne, C., 1987. Specific effect of guar gum or gum arabic on adaptation of cecal digestion to high fiber diets in the rat. J. Nutr. 117, 1556– 1561.
Walter, D.J., Eastwood, M.A., Brydon, W.G., Elton, R.A., 1988. Fermentation of wheat bran and gum arabic in rats fed on an elemental diet. Br. J. Nutr. 60, 225–232.
Yang, L., Pei, Z., 2006. Bacteria, inflammation, and colon cancer. World J. Gastroenterol. 12, 6741–6746.
Younes, H., Garleb, K., Behr, S., Remesy, C., Demigne, C., 1995. Fermentable fibers or oligosaccharides reduce urinary nitrogen excretion by increasing urea disposal in the rat cecum. J. Nutr. 125, 1010–1016.